Abstract
Dual antiplatelet therapy (DAPT) is required to prevent atherothrombotic events in patients with acute coronary syndrome (ACS) treated with percutaneous coronary intervention (PCI). The default DAPT strategy, namely a potent P2Y12 inhibitor combined with aspirin for 12 months, exposes many patients to excess bleeding risk. Over the past years, alternative antithrombotic regimens have been proposed to reduce bleeding (DAPT abbreviation or de-escalation) or ischaemic (prolonged dual antithrombotic therapy) events. Abbreviation or de-escalation of DAPT is supported by (i) multiple trials showing these strategies to significantly reduce bleeding, particularly for the 20–40% of patients classified as high bleeding risk (HBR); (ii) low prevalence of stent thrombosis and recurrent myocardial infarction beyond 1–3 months post-ACS with the latest generation of drug-eluting stents, and (iii) the recognition that HBR is far more prevalent than high ischaemic risk. Amongst patients at HBR, standard DAPT, in comparison to abbreviated or de-escalated DAPT, increases the net risk of major adverse events, even in the presence of high ischaemic risk. Conversely, amongst patients at high ischaemic risk, without HBR, prolonged dual antithrombotic therapy reduces longer-term thrombotic risk. Recognizing risk factors and assessing the magnitude of bleeding and ischaemic risks are essential. Since there are no ideal scoring systems to balance ischaemic and bleeding risks, and many overlap, focus should be on managing the risk most amenable to modification, namely bleeding, which should dominate the decision-making over ischaemic risk when choosing a DAPT regimen. This document provides practical advice regarding best practice for personalizing DAPT in patients with ACS undergoing PCI, with evidence-based clinical consensus statements on selecting the most appropriate antiplatelet strategy to optimize clinical outcomes.
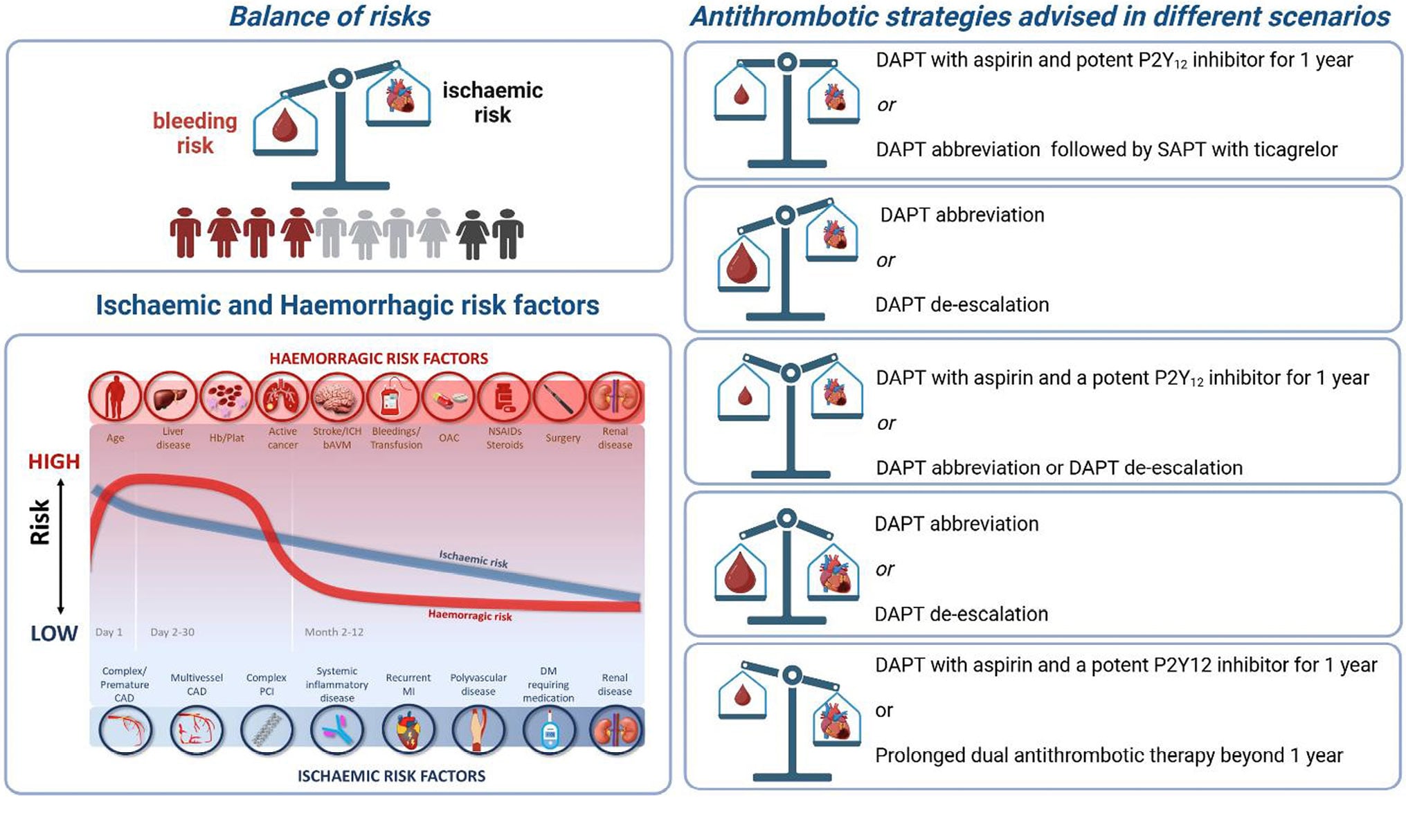
Graphical abstract.
Inroduction
The 2023 European Society of Cardiology (ESC) Guidelines for the Management of Acute Coronary Syndromes (ACS) recommend that the default dual antiplatelet therapy (DAPT) regimen should consist of a potent P2Y12 receptor inhibitor (prasugrel or ticagrelor) combined with aspirin for 12 months.1 The guidelines state that, in specific clinical scenarios, this default DAPT can be shortened (<12 months), extended (>12 months), or modified (DAPT de-escalation). There is, however, a need in the scientific community for practical guidance regarding the phenotype of patients most suited to these alternative antiplatelet regimens. The theme of the ESC 2024 congress in London was ‘Personalizing Cardiovascular Care’, devoted to improving patient outcomes. The current consensus document, from the ESC Working Group on Thrombosis together with the Association for Acute Cardiovascular Care (ACVC), the European Association of Percutaneous Cardiovascular Interventions (EAPCI) and the ESC Working Group on Cardiovascular Pharmacotherapy sets out to complement the ESC guidelines, providing practical advice on weighing up ischaemic and bleeding risks, in-depth review of available trial data, and evidence-based clinical consensus statements to aid clinicians in selecting the most appropriate antiplatelet strategy for their ACS patients, to optimize clinical outcomes.
Methodology
Definitions
Published some months before the ESC guidelines, the Academic Research Consortium defined de-escalation of DAPT in ACS patients as the ‘umbrella’ term covering strategies to reduce the antiplatelet effect by means of changes in the type, dose, or number of drugs used to inhibit platelets.2 In this document, we adhere to the terminology used in the ESC guidelines, namely the terms ‘shortened’ or ‘abbreviated’ DAPT duration when <12 months of DAPT is given, and DAPT ‘de-escalation’ when, after a brief period of conventional DAPT, a less potent P2Y12 inhibitor is used as part of DAPT. The term ‘standard DAPT’ will refer to the combination of aspirin with either ticagrelor or prasugrel given for 1-year post-ACS.
Literature search and consensus
Relevant clinical trials in patients with ACS treated with percutaneous coronary intervention (PCI) were identified from the literature, searching PubMed, Embase, and Cochrane Library databases to November 2025, with no restriction on language. Reference lists of selected papers were also checked for additional papers. All authors reviewed all sections of the manuscript and evaluated evidence according to the current ESC scientific document policy,3 with the strength of advice for the consensus statements based on evidence and consensus of the writing group (Figure 1). A 2-round modified Delphi method was used to reach consensus. In iterative rounds, panellists replied to the questionnaire, and group responses were compiled and provided to the participants. The panellists then re-evaluated their initial answer after considering the responses of the other panellists. Consensus was defined as ≥85% of participants agreeing with the consensus statement. All participants approved the final statements.

Figure 1. Scale and symbols representing the strength of advice statements, based on evidence and consensus of the writing group.
Standard dual antiplatelet therapy treatment for acute coronary syndrome: review of the evidence
After an ACS, international guidelines recommend DAPT for 12 months, combining aspirin with a P2Y12 inhibitor (clopidogrel, prasugrel, or ticagrelor).14 The 12-month recommendation is mainly driven by the findings of the Clopidogrel in Unstable Angina to Prevent Recurrent Events (CURE) trial, which was performed with an earlier generation of stents, with much higher rates of stent thrombosis (ST) than currently seen (Table 1).
Ticagrelor and prasugrel are recommended according to the 2023 ESC guidelines for ACS management over clopidogrel,1 based on the results of the PLATelet inhibition and patient Outcomes (PLATO) and TRial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet InhibitioN with Prasugrel Thrombolysis In Myocardial Infarction 38 (TRITON-TIMI 38) studies, which showed the superiority of ticagrelor and prasugrel over clopidogrel, respectively56 Ticagrelor reduced cardiovascular death and re-infarction compared to clopidogrel, while prasugrel reduced re-infarction alone. Both ticagrelor and prasugrel increased bleeding compared to clopidogrel. Subsequently, the open-label Intracoronary Stenting and Antithrombotic Regimen Rapid Early Action for Coronary Treatment (ISAR-REACT) 5 randomized controlled trial (RCT) compared 12 months’ DAPT including prasugrel vs. ticagrelor and showed that prasugrel reduced the risk of the composite of death, myocardial infarction, or stroke without an increase in bleeding complications,7 although some issues about the study design (e.g. pre-treatment with ticagrelor in non-ST-elevation ACS patients vs. administration of prasugrel after coronary angiography, open label) led to debate about the weight of the results.8 Current guidelines recommend that clopidogrel should be reserved for patients at high bleeding risk (HBR) or when used in combination with oral anticoagulation (OAC).19
GT 01
Table 1. Main randomized clinical trials informing standard duration of DAPT in patients with ACS.
| Study (year) | n | Treatment groups | Primary endpoint | Main result | Other relevant efficacy or safety endpoints |
|---|---|---|---|---|---|
| CURE (2001) | 12 562 | Aspirin vs. Aspirin + clopidogrel (loading dose 300 and 75 mg daily) | Death from CV causes, non-fatal MI, or stroke at 1 year |
Clopidogrel reduced the primary endpoint compared to placebo (9.3% vs. 11.4%; RR 0.80; 95%CI 0.72–0.90; P<0.001). | Increased major bleeding in the clopidogrel group (3.7% vs. 2.7%; RR 1.38; P=0.001). No increase in life-threatening bleeding or haemorrhagic strokes with clopidogrel Refractory ischemia during initial hospitalization was reduced by clopidogrel, as well as myocardial infarction (Q wave) |
| CURRENT-OASIS 7 (2010) | 25 086 | 2 × 2 factorial design Clopidogrel 600 mg LD + 150 mg for 6 days vs. Clopidogrel 300 LD + 75 mg Aspirin 300–375 mg daily vs. Aspirin 75– 100 mg daily |
CV death, MI, or stroke at 30 days | No significant difference between higher and lower dose of clopidogrel (4.2% vs. 4.4%; HR 0.94; 95%CI 0.83–1.06; P=0.30) or between higher and lower dose of aspirin (4.2% vs. 4.4%; HR 0.97, 95%CI 0.86–1.09; P =0.61). | No differences in major bleeding between aspirin doses Increased rates of major bleeding with double-dose clopidogrel compared to standard-dose (2.5% vs. 2.0%; HR 1.24; 95%CI 1.05–1.46; P=0.01) Double-dose clopidogrel reduced stent-thrombosis (1.6% vs. 2.3%; HR 0.68; 95%CI 0.55–0.85; P=0.001) |
| PLATO (2009) | 18 624 | Aspirin + clopidogrel vs. Aspirin + ticagrelor (loading dose 180 mg followed by 90 mg bid) | CV death, MI, or stroke at 1 year |
Ticagrelor reduced the primary endpoint compared to clopidogrel (9.8% vs. 11.7%; HR 0.84, 95%CI 0.77–0.92; P<0.001) | Ticagrelor increased fatal intracranial bleeding and major bleeding not related to CABG (4.5% vs. 3.8%, P=0.03) Ticagrelor reduced myocardial infarction, death from vascular causes (4.0% vs. 5.1%; P=0.001) and all-cause death (4.5% vs. 5.9%; P<0.001). |
| TRITON-TIMI 38 (2007) | 13 608 | Aspirin + clopidogrel vs. Aspirin + prasugrel (loading dose 60 mg, followed by 10 mg daily) | CV death, MI, or stroke at 15 months | Prasugrel reduced the primary endpoint compared to clopidogrel (9.9% vs. 12.1%; HR 0.81, 95%CI 0.73–0.90; P <0.001) | Prasugrel therapy was associated with increased rates of TIMI major bleeding compared to clopidogrel (2.4% vs. 1.1%; HR 1.32, 95%CI 1.03–1.46; P=0.03) Prasugrel reduced the rates of myocardial infarction, urgent target-vessel revascularization and stent thrombosis (2.4% vs. 1.1%; P <0.001) |
| ISAR REACT-5 (2019) | 4018 | Aspirin + ticagrelor vs. Aspirin + prasugrel | Death, MI, or stroke at 1 year |
Prasugrel reduced the primary endpoint compared to ticagrelor (9.3% vs. 6.9%; HR 1.36; 95%CI 1.09–1.70; P=0.006). | No significant differences were observed in BARC major bleeding (safety endpoint) between drugs (5.4% vs. 4.8%; HR 1.12; 95%CI 0.83–1.51; P=0.46) No significant differences were seen in death or in definite or probable stent thrombosis |
| ACS, acute coronary syndrome; BARC, bleeding academic research consortium; CABG, coronary artery bypass graft; CI, confidence interval; CV, cardiovascular; HR, hazard ratio; MI, myocardial infarction; NSTEACS, non-ST-elevation acute coronary syndrome; RR, relative risk; TIMI, thrombolysis in myocardial infarction. | |||||

GT 01.
Assessing ischaemic risk
Incidence of ischaemic events with standard dual antiplatelet therapy
Ischaemic events following an ACS with standard DAPT occur in 5–10% of patients in the first year.710 In ACS patients treated with the current generation of drug-eluting stents with thin struts, the risk of ST is low, occurring in 1–2% of patients in the first year, with ~70% of these occurring within the first 30 days (Figure 2).11121314
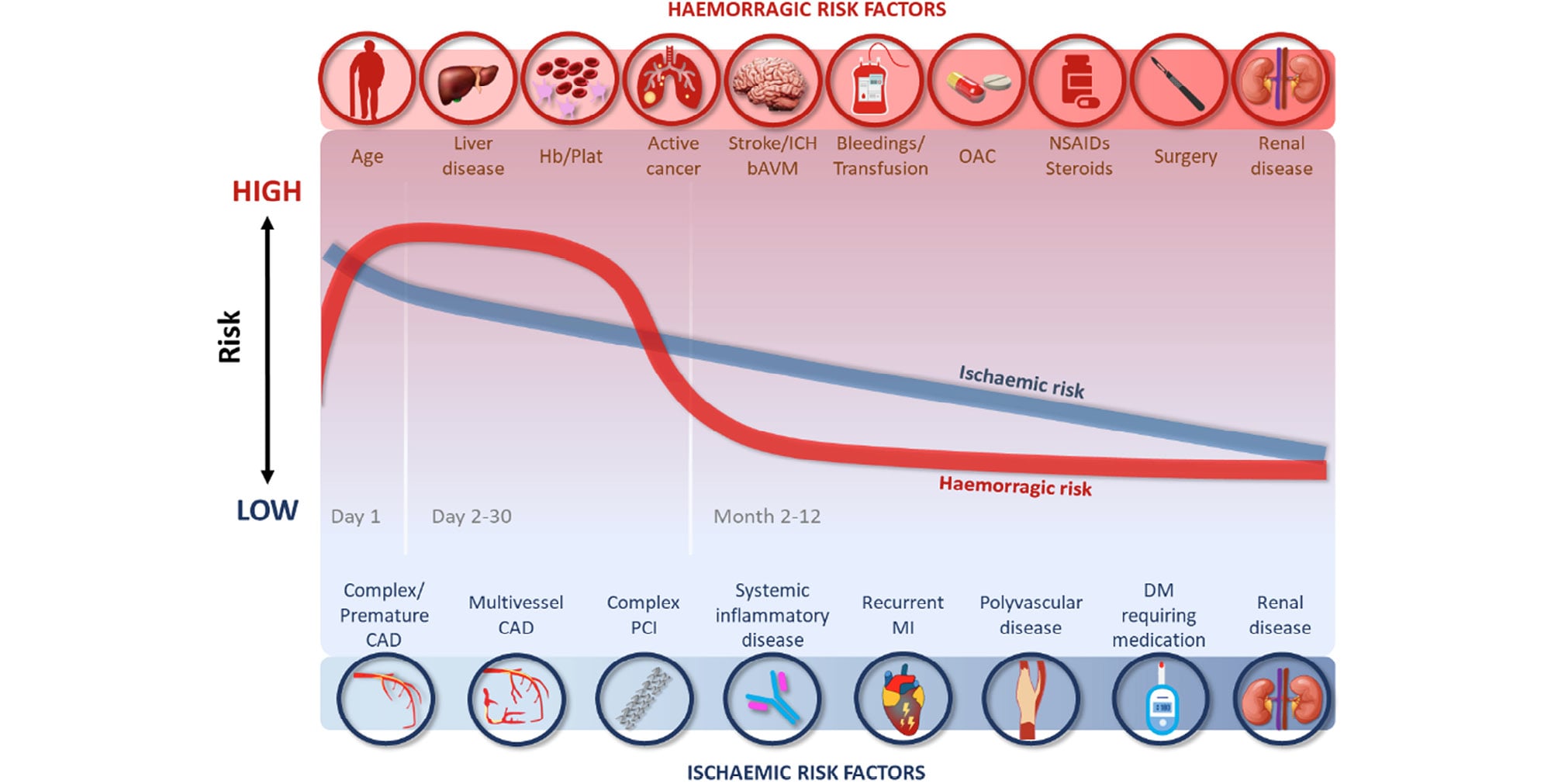
Figure 2. Risk of ischaemic and bleeding events over time after an ACS. ACS, acute coronary syndrome; bAVM, brain arteriovenous malformation; CAD, coronary artery disease; DM, diabetes mellitus; Hb/Plat, anaemia or platelet disorders; ICH, intracranial haemorrhage; MI, myocardial infarction; NSAIDs, non-steroidal anti-inflammatory drugs; OAC, oral anticoagulation; PCI, percutaneous coronary intervention.
Clinical, anatomical, or procedural risk factors for ischaemic events
Several studies identified common clinical risk factors for recurrent ischaemia (Figure 2), including age, diabetes mellitus, history of recurrent myocardial infarction, multivessel coronary artery disease, premature (<45 years) or accelerated (new lesion within a 2-year time frame) coronary artery disease, complexity of PCI, prior ST, prior stroke, concomitant systemic inflammatory disease (e.g. human immunodeficiency virus, systemic lupus erythematosus, chronic arthritis), polyvascular disease (coronary and peripheral artery disease) or chronic kidney disease (CKD).11516 The anatomical/periprocedural factors that have been shown to be associated with ischaemic events are the implantation of multiple stents (≥3), multiple lesions treated (≥3), total stent length >60 mm, and complexity of revascularization (left main, bifurcation stenting with ≥2 stents, chronic total occlusion, stenting of last patent vessel, history of ST on antiplatelet treatment).1718 However, many ischaemic risk factors are also risk factors for bleeding.19 In the ARC–High Bleeding Risk trade-off model, several variables were associated with increased risk of both ischaemic and bleeding events, including anaemia, CKD, current smoking, and a complex PCI procedure (the latter associated with bleeding likely through use of additional antithrombotic agents). Four variables were associated with ischaemic risk only: prior myocardial infarction, diabetes mellitus, ST-segment elevation, or non–ST-segment elevation myocardial infarction at presentation, and the use of bare metal stents.19
Biomarkers of ischaemic risk
Several platelet function tests (PFT) are available to measure high on-treatment platelet reactivity. While studies showed a strong and consistent relationship between high on-treatment platelet reactivity and increased ischaemic risk, there have been no studies to date to show that adjusting antiplatelet medication based on the results of PFT can improve prognosis.20 For this reason, and lack of routine availability, their current clinical application is limited. Other conventional tests of ischaemic risk include C-reactive protein (CRP), D-dimer, fibrinogen, and troponin. Among them, the most studied is CRP, which also has the most consistent association with ischaemic complications.2122
Novel biomarkers in the research pipeline, but not routinely available, include markers of inflammation and oxidative stress (miRNAs, interleukin-6, myeloperoxidase, galectin-3, endocan, soluble lectin-like oxidized low-density lipoprotein receptor-1, procalcitonin, etc.), fibrosis (galectin-3, growth differentiating factor [GDF]-15, soluble Suppression of Tumorigenicity [sST2], etc.), neurohumoral activation [copeptin, brain natriuretic peptide (BNP) and n-terminal pro-BNP, mid-regional proadrenomedullin, etc.],2324 and platelet activation (P-selectin, von Willebrand factor, platelet-derived growth factor A).25
Ischaemic risk scores
Various risk scores such as the Thrombolysis in Myocardial Infarction (TIMI) risk score, the GRACE 3.0 score and the HEART score have been developed to predict 14-day to 6-month clinical outcomes in ACS patients.262728 While scores such as GRACE are able to identify which patients benefit from early intervention, their use has not been shown to improve clinical outcomes,2930 nor have the scores been developed to guide antithrombotic therapy strategy. The ABC-ACS ischaemia score has shown improved discrimination for predicting 1-year cardiovascular death or myocardial infarction, but requires measurement of plasma GDF-15 as well as NT-proBNP.31 The PREdicting bleeding Complications In patients undergoing Stent implantation and subsEquent Dual Anti Platelet Therapy (PRECISE-DAPT) score may be used at patient discharge to predict the balance of ischaemic and bleeding risk, in order to guide the initial duration of DAPT.32 A PRECISE-DAPT score <25 identifies patients in whom prolonged DAPT reduces the risk of ischaemic events, without this benefit being offset by increased bleeding. There are no clinical trials evaluating clinical risk scores to guide the use of DAPT, and use of scores has not been shown to improve clinical outcomes.
GT 02

GT 02.
Assessing bleeding risk
Incidence of haemorrhagic events with standard dual antiplatelet therapy
The incidence of bleeding with DAPT is dependent on the definition of bleeding, the type and dose of P2Y12 inhibitor used (higher bleeding risk with the more potent P2Y12 inhibitors), and the bleeding risk category of the patient. In clinical trials of DAPT in patients with ACS, the incidence of major bleeding unrelated to coronary artery bypass grafting (CABG) ranged from 2% to 5% for the period of DAPT continuation,56733 and was similar in observational trials over a 1-year follow-up.3435 When considering both major and minor bleeding, a 12-month incidence of up to 35–40% has been reported, but still varying with the classification used.34 In patients treated with PCI, the risk of bleeding is highest in the acute phase and then gradually falls (Figure 2).32 Following discharge, the incidence of bleeding is highest in the first weeks, with minor bleeding most commonly reported.34
Clinical, anatomical, or procedural risk factors for bleeding
Frailty, older age, female gender, and low body weight are established predictors of increased bleeding risk.3637 Additionally, CKD is an independent risk factor for bleeding after PCI, with risk increasing incrementally with worsening kidney function,373839 such that severe or end-stage CKD [estimated glomerular filtration rate (eGFR) <30 mL/min] is a major risk factor, while moderate disease (eGFR 30–59 mL/min) is a minor bleeding risk factor.37 Additional risk factors include a history of bleeding, as well as the presence of anaemia, thrombocytopenia, leucocytosis, or concomitant OAC therapy.10323637 PCI-related factors associated with increased bleeding risk include the use of femoral, compared to radial approach.404142 Use of adjunctive antithrombotic therapy during PCI, such as glycoprotein IIb/IIIa inhibitors, and administration of non-weight-adjusted unfractionated heparin, particularly when coupled with femoral access, increase the risk of haemorrhagic complications.3643 Mitigating any modifiable bleeding risk factors (such as the avoidance of femoral access and adjunctive antithrombotic agents) can significantly reduce haemorrhagic complications.
Biomarkers of bleeding risk
Identification of better biomarkers of bleeding is needed to identify patients at HBR who may benefit from closer monitoring and personalized measures to reduce bleeding complications. The PLATO trial showed a strong association between GDF-15 and major non-CABG-related (including fatal) bleeding at ACS presentation44; an effect that persisted up to 1-month.4546 An elevated white cell count on admission has been proposed as an independent predictor of 30-day major bleeding.47 More recently, red cell distribution width, a measure of the range of variation of red blood cell volume, as well as aspartate aminotransferase levels, were significantly associated with bleeding in patients on DAPT post-PCI, and the addition of these to the DAPT score improved the prediction of bleeding over the DAPT score alone.48
Bleeding risk scores
Various bleeding risk scores have been evaluated in patients with ACS.32495051 A meta-analysis including studies incorporating the DAPT score, which may help uncouple bleeding and ischaemic risk following PCI, showed that patients with a DAPT score ≥2 had an increased ischaemic risk and reduced bleeding risk compared to those with lower scores.49 In a systematic review and large meta-analysis, patients with a PRECISE-DAPT score ≥25 experienced a significantly higher rate of any out-of-hospital bleeding and major bleeding,52 with the score showing only moderate discriminative power in predicting major bleeding events at 1 year.
In the 2020 ESC Guidelines for the management of ACS in patients presenting without persistent ST-segment elevation,53 various bleeding risk scores are discussed, but it is noted that none of these risk prediction models have been prospectively tested in large RCTs, and therefore, their value in improving patient outcomes remains unclear. Since then, the Academic Research Consortium HBR score identified 20 clinical criteria as major or minor risk factors for Bleeding Academic Research Consortium [BARC] types 3–5 bleeding post-PCI, with HBR defined in binary terms when at least 1 major or 2 minor criteria were present.37 In a validation study of 6641 ARC-HBR patients undergoing PCI, 5% sustained myocardial infarction/ST, and 6% experienced BARC 3–5 bleeding.19 There were eight predictors of myocardial infarction and/or ST and eight of major bleeding, four of which predicted both, demonstrating a moderate discriminative ability of the ARC-HBR criteria.19 The moderate discriminative ability of all available risk scores highlights the need for future improved risk prediction tools in the field.
GT 03

GT 03.
Combined evaluation of bleeding and ischaemic risks: change over time following acute coronary syndrome
Factors influencing bleeding and ischaemic risk in patients with ACS intertwine both during the acute phase and during long-term secondary prevention (Figure 2). A subanalysis of the HORIZONS-AMI (Harmonizing Outcomes with Revascularization and Stents in Acute Myocardial Infarction) trial aimed to define the average daily ischaemic and bleeding rates within the first year after primary PCI for STEMI.54 Both ischaemic (defined as cardiac death, re-infarction, and ST) as well as bleeding (defined as TIMI major and minor bleeding) events were shown to decline considerably after the acute phase of myocardial infarction but with different trends. Within the first 24 h (acute phase) of the index event, bleeding events tend to increase, while the ischaemic risk slowly decreases.54 In the acute phase, bleeding events are more frequent in patients treated with an interventional approach than in those managed conservatively, driven by the use of intra-procedural drugs (anticoagulation or antiplatelet agents) and access site complications (more frequent with femoral compared to radial access).55 In the early phase following primary PCI (Days 1–30), both bleeding and ischaemic risks decline, with bleeding risk decreasing more rapidly than ischaemic risk.1 The difference in ischaemic and bleeding risk in any individual justifies a personalized antiplatelet strategy post-ACS.
Therapeutic options to minimize bleeding risk
Antithrombotic regimens that can be used as an alternative to standard DAPT to reduce bleeding can be grouped into two main categories, namely abbreviation or de-escalation of DAPT (Figure 3). Of note, there has been no direct head-to-head comparison of these two regimens performed to date.
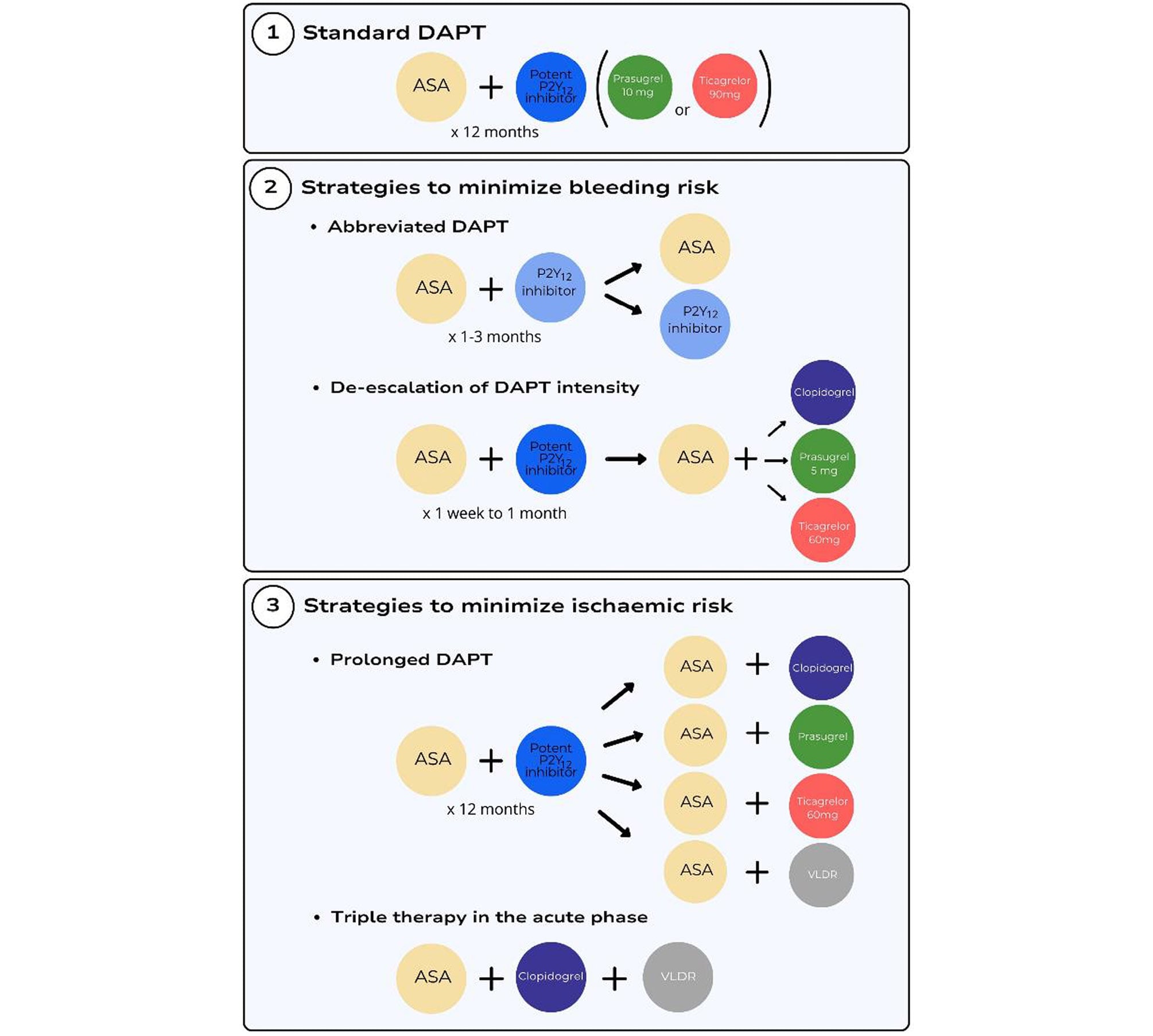
Figure 3. Antithrombotic therapy strategies after an ACS. ASA, aspirin; DAPT, dual antiplatelet therapy; VLDR, very low dose rivaroxaban.
Differences between P2Y12 inhibitors with respect to bleeding risk
Adding a P2Y12 inhibitor to aspirin increases the risk of bleeding, which is higher with prasugrel or ticagrelor than with clopidogrel. In the CURE trial, GUSTO major bleeding was increased with clopidogrel compared to placebo (3.7% vs. 2.7%; P=0.001).33 In TRITON-TIMI 38, TIMI major bleeding (2.4% vs. 1.8%, P=0.03), life-threatening bleeding (1.4% vs. 0.9%; P=0.01), and fatal bleeding (0.4% vs. 0.1%; P=0.002) were increased with prasugrel compared to clopidogrel.6 In the PLATO trial, ticagrelor increased non-CABG-related TIMI major bleeding (2.8% vs. 2.2%; P=0.03), including more fatal intracranial bleeding.5
Abbreviated dual antiplatelet therapy
Followed by aspirin monotherapy
Thirteen RCTs have investigated the comparative effectiveness and safety of an abbreviated DAPT regimen (ranging from 1 to 6 months), followed by aspirin monotherapy, compared with standard DAPT after PCI, with the timing of the primary endpoint ranging from 12 to 18 months (Table 2). Although many of these trials included only a small subgroup of patients with ACS, such abbreviated DAPT followed by aspirin monotherapy appeared to consistently reduce bleeding risk.56575859606162636465666768
Table 2. Main randomized clinical trials evaluating a strategy of abbreviation of DAPT followed by aspirin monotherapy in patients with ACS.
| Study (year) | n | Ethnicity | Treatment groups | Primary endpoint | Main result | Other relevant efficacy or safety endpoints | Comments |
|---|---|---|---|---|---|---|---|
| RESET (2012) | 2117 | East-Asian (Korean) 100% | 3 months DAPT (aspirin + clopidogrel) vs. 12 months DAPT (aspirin + clopidogrel) |
CV death, MI, ST, target/vessel revascularization, or bleeding at 1 year | Abbreviated DAPT achieved non-inferiority for the primary endpoint (4.7% vs. 4.7%; difference 0.0%; 95%CI −2.5% to 2.5%; P for non-inferiority <0.001; P for superiority = 0.84) | No significant difference in major bleeding (0.2% vs. 0.6%; difference −0.4%; 95%CI −0.9% to 0.1%; P=0.16) | Lower than expected event rates. Potent P2Y12 inhibitors not used. |
| EXCELLENT (2012) | 1443 | East-Asian (Korean) 100% | 6 months DAPT (aspirin + clopidogrel) vs. 12 months DAPT (aspirin + clopidogrel) | Cardiac death, MI, or ischaemia driven target revascularization at 12 months | Abbreviated DAPT was non-inferior with respect to the primary endpoint (4.8% vs. 4.3%; HR 1.14; 95%CI 0.70–1.86; f for non-inferiority = 0.001; P for superiority = 0.60) | No significant difference in TIMI major bleeding (0.3% vs. 0.6%; HR 0.50; 95% CI 0.09–2.73; P=0.64) Numerical trend towards increased ST in the abbreviated DAPT arm. |
Wide margin of non-inferiority. Lower than expected event rates. Potent P2Y12 inhibitors not used. |
| OPTIMIZE (2013) | 3211 | Enrolment in South-America (Brazil) 100% | 3 months DAPT (aspirin + clopidogrel) vs. 12 months DAPT (aspirin + clopidogrel) | All-cause death, MI, stroke, or major bleeding (NACE) at 12 months | Abbreviated DAPT was non-inferior with respect to the primary endpoint (6.0% vs. 5.8%; HR 1.03; 95%CI 0.77–1.38; P for non-inferiority = 0.002) | No significant differences in major bleeding (0.6% vs. 0.9%; HR 0.71; 95%CI 0.32–1.60) or MACE | Low risk patients and lower than expected event rates Potent P2Y12 inhibitors not used. |
| SECURITY (2014) | 1399 | Enrolment in Europe 100% | 6 months DAPT (aspirin + clopidogrel) vs. 12 months DAPT (aspirin + clopidogrel) | Cardiac death, MI, stroke, definite or probable ST, or BARC ≥ 3 bleeding (NACE) at 12 months. | Abbreviated DAPT met non-inferiority criteria for the primary endpoint (4.5% vs. 3.7%; difference 0.8%; 95%CI −2.4% to 1.7%; P for non-inferiority <0.5). | No significant difference in the main secondary endpoint of cardiac death, MI, stroke, definite or probable ST, or BARC ≥ 2 bleeding was observed. | Minimal use of P2Y12 inhibitors others than clopidogrel Low risk patients and lower than expected event rates Prematurely stopped due to low enrolment and no differences for the primary endpoint |
| ITALIC (2015) | 2031 | Inclusion in Europe (52 centres) and Middle East (5 centres), % not available | 6 months DAPT (aspirin + clopidogrel) vs. 24 months DAPT (aspirin + clopidogrel) | Death, MI, urgent target vessel revascularization, stroke, and major bleeding at 12 months. | Abbreviated DAPT was non-inferior with respect to the primary endpoint (1.6% vs. 1.5%; HR 1.072; 95%CI 0.517–2.221; P for non-inferiority = 0.0002) | No significant differences in secondary ischaemic or bleeding endpoints were found. | Patients not randomized if poor-responsiveness to aspirin Minimal use of P2Y12 inhibitors others than clopidogrel Prematurely stopped due to low recruitment problems |
| I-LOVE-IT 2 (2016) | 1829 | East-Asian (Chinese) 100% | 6 months DAPT (aspirin + clopidogrel) vs. 12 months DAPT (aspirin + clopidogrel) | Cardiac death, target vessel MI, or clinically driven target lesion revascularization at 12 months | Abbreviated DAPT reached non-inferiority for the primary endpoint (6.8% vs. 5.9%; difference 0.87%; 95%CI −1.37% to 3.11%; P for non-inferiority = 0.0065). | No significant difference in the composite of all-cause death, all-cause MI, stroke, or BARC ≥3 bleeding (7.2% vs. 6.4%; P= 0.53) or in BARC ≥3 bleeding (1.2% vs. 0.7%; P = 0.21). | Substudy with low event rates. Potent P2Y12 inhibitors not used. |
| IVUS-XPL (2016) | 1400 | East-Asian (Korean) 100% | 6 months DAPT (aspirin + clopidogrel) vs. 12 months DAPT (aspirin + clopidogrel) | Cardiac death, MI, stroke, definite or probable ST, or TIMI major bleeding (NACE) at 1 year | No difference between abbreviated and standard DAPT duration for the primary endpoint (2.2% vs. 2.1%; HR 1.07; 95%CI 0.52–2.22; P=0.854) | No significant differences in secondary ischaemic or bleeding endpoints were observed. | Potent P2Y12 inhibitors not used. Low risk patients |
| NIPPON (2017) | 3773 | East-Asian (Japanese) 100% | 6 months DAPT (aspirin + clopidogrel) vs. 18 months DAPT (aspirin + clopidogrel) | All-cause mortality, MI, stroke, and major bleeding (NACE) from 6 to 18 months | Abbreviated DAPT achieved non-inferiority for the primary endpoint (2.1% vs. 1.5%; difference −0.6%; 95%CI −1.5% to 0.3%; P for superiority =0.024). | No significant differences in secondary ischaemic or bleeding endpoints were observed. | Minimal use of P2Y12 inhibitors others than clopidogrel Low risk patients and lower than expected event rates |
| DAPT-STEMI (2018) | 870 | Western (European), % not available | 6 months DAPT (aspirin + P2Y12 inhibitor) vs. 12 months DAPT (aspirin + P2Y12 inhibitor) | All-cause mortality, MI, any revascularization, stroke, and TIMI major bleeding (NACE) at 18 months after randomization. | Abbreviated DAPT was non-inferior with respect to the primary endpoint (4.8% vs. 6.6%; HR 0.73, 95%CI 0.41–1.27; P for non-inferiority = 0.004; P for superiority 0.26). | No significant difference in TIMI major bleeding (0.2% vs. 0.5%; HR 0.51; 95%CI 0.05–5.57; P=0.49). | STEMI patients with primary PCI randomized if event-free at 6 months Small sample size and lower than expected event rates |
| OPTIMA-C (2018) | 1368 | East-Asian (Korean) 100% | 6 months DAPT (aspirin + clopidogrel) vs. 12 months DAPT (aspirin + clopidogrel) | Cardiac death, MI, or ischaemia-driven target lesion revascularization at 12 months. | Abbreviated DAPT was non-inferior with respect to the primary endpoint (1.2% vs. 0.6%; difference 0.6%; 95%CI −0.4% to 1.6%; P for non-inferiority <0.05; P for superiority = 0.24). | No TIMI major bleeding events in each arm | Very low risk population. Potent P2Y12 inhibitors not used. |
| SMART DATE (2018) | 2712 | East-Asian (Korean) 100% | 6 months DAPT (aspirin + P2Y12 inhibitor) vs. 12 months DAPT (aspirin + P2Y12 inhibitor) | All-cause death, MI, or stroke at 18 months. | Non-inferiority of abbreviated DAPT for the primary endpoint (4.7% vs. 4.2%; HR 1.13; 95% CI 0.79–1.62; P for non-inferiority = 0.03; P for superiority = 0.51). | Significant increase in the rates of MI with abbreviated DAPT (1.8% vs. 0.8%; HR 2.41; 95% CI 1.15–5.05; P=0.02). No significant difference in BARC ≥2 bleeding (2.7% vs. 3.9%; HR 0.69; 95%CI 0.45–1.05; P=0·09). | Wide non-inferiority margin Approximately 80% use of clopidogrel as part of DAPT |
| REDUCE (2019) | 1496 | Caucasian 68.6% Asian 30.5% Black 0.5% Other 0.4% | 3 months DAPT (aspirin + P2Y12 inhibitor) vs. 12 months DAPT (aspirin + P2Y12 inhibitor) | All-cause mortality, MI, ST, stroke, target vessel revascularization, or BARC ≥ 2 bleeding (NACE) at 12 months. | Abbreviated DAPT was non-inferior with respect to the primary endpoint (8.2% vs. 8.4%; HR 0.97; 95%CI 0.68–1.39; P for non-inferiority <0.001; P for superiority 0.80). | No significant difference in BARC ≥2 (2.5% vs. 3.0%; HR 0.83; 95%CI 0.45–1.55; P=0.540). Numerically higher rates of mortality and ST in the abbreviated DAPT group. | Open label and non-inferiority design with wide margin. Lower than expected event rates. |
| One-Month DAPT (2021) | 3020 | East-Asian (Korean) 100% | 1 months DAPT (aspirin + P2Y12 inhibitor) vs. 6–12 months DAPT (aspirin + P2Y12 inhibitor) | Cardiac death, non-fatal MI, target vessel revascularization, stroke, or major bleeding (NACE) at 1 year | Non-inferiority of abbreviated DAPT for the primary endpoint (5.9% vs. 6.5%; HR 0.90; 95%CI 0.68–1.20; P for non-inferiority <0.001; P for superiority =0.475). | No significant differences in major bleeding (1.7% vs. 2.5%; HR 0.69; 95%CI 0.42–1.13; P=0.136) or in ischaemic outcomes. | Clopidogrel in the majority of patients (94%) Inclusion only if PCI of non-complex lesions. |
| ACS, acute coronary syndrome; BARC, bleeding academic research consortium; CI, confidence interval; CV, cardiovascular; DAPT, dual antiplatelet therapy; GUSTO, global use of strategies to open occluded coronary arteries; HPR, high on-treatment platelet reactivity; HR, hazard ratio; LPR, low on-treatment platelet reactivity; MACE, major adverse cardiovascular events; MI, myocardial infarction; NACE, net adverse clinical events; SAPT, single antiplatelet therapy ST, stent thrombosis; STEMI, ST-elevation myocardial infarction; TIMI, thrombolysis in myocardial infarction. | |||||||
Followed by P2Y12 inhibitor monotherapy
Twelve RCTs have so far assessed the comparative effectiveness and safety of a short DAPT regimen (median of 16 days to maximum 3 months), followed by P2Y12 inhibitor monotherapy, compared with standard DAPT in ACS patients (Table 3),697071727374757677787980 which includes one recent trial that has evaluated this strategy using personalized risk stratification to allocate DAPT duration to patients.77
Aspirin withdrawal has been consistently associated with a lower risk of major bleeding compared with 12 months DAPT. The Single Versus Dual Antiplatelet Therapy (Sidney-2) Collaboration was an individual patient data meta-analysis of six studies involving some 13 750 ACS patients, comparing 1–3 months of DAPT followed by ticagrelor, clopidogrel, or prasugrel in 77%, 22.2%, and 0.8% of the patients, respectively. Monotherapy with a P2Y12 inhibitor was associated with 60% risk reduction in BARC 3 or 5 bleeding compared to DAPT, with the number needed to treat for benefit of 71.81 This benefit was not confined to patients with HBR features and proved highly consistent across all explored patient subgroups. However, the timing of aspirin withdrawal may be critical. In STOPDAPT-3, patients (75% had ACS) were randomized just before PCI to either low-dose prasugrel (3.75 mg/day) monotherapy or to DAPT with aspirin and prasugrel (3.75 mg/ day).82 At 1 month, bleeding was similar in the two treatment arms, but while prasugrel monotherapy was non-inferior to DAPT for cardiovascular events, prasugrel monotherapy was associated with a signal for excess ischaemic risk.82 The NEOMINDSET trial evaluated the strategy of very early aspirin withdrawal by randomizing patients within 4 days of ACS hospitalization to aspirin withdrawal while continuing potent P2Y12 inhibitor monotherapy (ticagrelor or prasugrel) or to DAPT for 12 months.79 Although early aspirin withdrawal reduced major or clinically relevant non-major bleeding, it was not non-inferior to 12 months’ DAPT, with an increase in death and ischaemic events, including ST.
Current evidence also suggests that the protection from ischaemic events with P2Y12 inhibitor monotherapy compared with DAPT may differ, based on the type of P2Y12 inhibitor used as monotherapy. Studies investigating ticagrelor monotherapy compared with DAPT in ACS patients did not raise any concerns regarding the risk overall of cardiovascular death, myocardial infarction, stroke, or ST. Individual patient data or aggregate data meta-analyses provided evidence for a reduced mortality risk with ticagrelor monotherapy compared with DAPT, comprising aspirin and clopidogrel.83 However, these findings should be interpreted with caution, since no direct comparisons between different P2Y12 inhibitor monotherapy options as part of a de-escalation are available. In a trial involving 4169 patients with ACS comparing standard DAPT with clopidogrel monotherapy after 1-month DAPT, the composite of cardiovascular death, myocardial infarction, ST, and stroke exceeded the pre-defined non-inferiority boundaries at 1-year due to a significant excess of myocardial infarction in the clopidogrel monotherapy arm.74 The evidence with prasugrel monotherapy is limited and inconclusive, with the few available trials assessing low, unconventional doses.7882
Table 3. Main randomized clinical trials evaluating a strategy of abbreviation of DAPT followed by P2Y12 monotherapy in patients with ACS.
| Study (year) | n | Ethnicity | Treatment groups | Primary endpoint | Main result | Other relevant efficacy or safety endpoints | Comments |
|---|---|---|---|---|---|---|---|
| GLOBAL LEADERS ACS subgroup (2018) | 7487 | Enrolment in the overall trial: Western Europe 77.2% Eastern Europe 18.8% Other 3.9% | 1 month DAPT (aspirin + ticagrelor) followed by 23 months ticagrelor vs. 12 months DAPT (aspirin + ticagrelor) followed by aspirin monotherapy | All-cause mortality, or non-fatal new Q-wave MI at 2 years | Abbreviated DAPT was not superior with respect to the primary endpoint (3.92% vs. 4.52%; HR 0.86; 95%CI 0.69–1.08; P=0.19). | Abbreviated DAPT reduced BARC ≥3 bleeding at 24 months (1.95% vs. 2.68%; RR 0.73; 95% CI 0.54–0.98; P=0.037) | Subgroup analysis. Lower than expected event rates. No central adjudication of events |
| SMART-CHOICE (2019) | 2993 | East-Asian (Korean) 100% | 3 months DAPT (aspirin + P2Y12 inhibitor) followed by 9 months P2Y12 monotherapy vs. 12 months aspirin + ticagrelor | All-cause death, MI, or stroke at 12 months | Abbreviated DAPT achieved non-inferiority for the primary endpoint (2.9% vs. 2.5%; difference 0.4%; 95%CI, −∞% to 1.3%; P for non-inferiority = 0.007). | Abbreviated DAPT reduced BARC ≥2 bleeding (2.0% vs. 3.4%; HR 0.58; 95% CI 0.36–0.92; P=0.02). | Wide non-inferiority margin. Approximately 77% use of clopidogrel. |
| TWILIGHT-ACS (2020) | 4614 | Non-white 37.4% Enrolment in Asia 30% | 3 months DAPT (aspirin + ticagrelor) followed by ticagrelor for 12 months vs. 15 months DAPT (aspirin + ticagrelor) | BARC ≥ 2 bleeding at 12 months after randomization | Superiority of abbreviated DAPT for the primary endpoint (3.6% vs. 7.6%; HR 0.47; 95%CI 0.36–0.61; P<0.001). | No differences between strategies in the combined key secondary endpoint of all-cause death, non-fatal MI, or non-fatal stroke at 12 months after randomization (4.3% vs. 4.4%; HR 0.97; 95%CI 0.74–1.28; P=0.84) Abbreviated DAPT reduced BARC ≥3 bleeding (0.8% vs. 2.1% HR 0.36; 95% CI 0.20–0.62; P<0.001) | Randomization of patients if event-free at 3 months after PCI Lower than expected rates of ischaemic events |
| TICO (2020) | 3056 | East-Asian (Korean) 100% | 3 months DAPT (aspirin + ticagrelor) followed by 9 months ticagrelor vs. 12 months DAPT (aspirin + ticagrelor) | Major TIMI bleeding, death, MI, ST, stroke, or target-vessel revascularization (NACE) at 12 months | Abbreviated DAPT significantly reduced the rates of the primary endpoint (3.9% vs. 5.9%; HR0.66; 95%CI 0.34–0.91; P=0.01). | Abbreviated DAPT reduced TIMI major bleeding (1.7% vs. 3.0%; HR 0.56; 95%CI 0.34–0.91; P=0.02). No differences between groups in MACE. | Lower than expected event rates. Patients at high risk of bleeding excluded. |
| MASTER DAPT (2021) | 4434 | Enrolment in the trial: Europe 83.8% Asia 12.7% Other 3.5% |
1 month DAPT (aspirin + P2Y12) followed by SAPT vs. 3–12 months DAPT (aspirin + P2Y12) | Three ranked primary outcomes: • NACE: all-cause death, MI, stroke, or BARC ≥ 3 bleeding. • MACE: all-cause death, MI, or stroke. • BARC ≥ 2 bleeding. | Abbreviated DAPT was non-inferior with respect to the primary endpoints of NACE (7.5% vs. 7.7%; HR 0.97; 95%CI 0.78–1.20; P for non-inferiority <0.001) and MACE (6.1% vs. 5.9%; HR 1.02; 95%CI 0.80–1.30; P for non-inferiority <0.001). Abbreviated DAPT significantly reduced BARC ≥2 bleeding (6.5% vs. 9.4%; HR 0.68; 95%CI 0.55–0.85; P <0.001) | No significant differences in bleeding according to TIMI or GUSTO definitions. | Patients randomized if event-free at 1 month after PCI Patients included if at high risk of bleeding (38% with indication for oral anticoagulation) SAPT in abbreviated DAPT arm with P2Y12 inhibitor in ~70% of subjects |
| STOPDAPT-2 ACS (2022) |
4169 | East-Asian (Japanese) 100% | 1–2 months DAPT (ASA + clopidogrel or prasugrel 3.75 mg) followed by clopidogrel until 1 year vs. 1–2 months DAPT (ASA + clopidogrel or prasugrel 3.75 mg) followed by DAPT (ASA + clopidogrel) until 1 year | CV death, MI, ischemic, or haemorrhagic stroke, definite ST, or major or minor bleeding (NACE) at 12 months | Abbreviated DAPT failed to reach non-inferiority for the primary endpoint (3.2% vs. 2.8%; HR 1.14; 95%CI 0.80–1.62; P for non-inferiority = 0.06). | Increased rates of MI with abbreviated DAPT (1.59% vs. 0.85%; HR 1.91; 95%CI 1.06– 3.44). Abbreviated DAPT reduced TIMI major/ minor bleeding (1.17% vs. 0.54%; HR 0.46; 95% CI 0.23–0.94). |
Lower than expected event rates. |
| T-PASS (2024) | 2850 | East-Asian (Korean) 100% | <1 month DAPT (aspirin + ticagrelor) followed by 11 months ticagrelor vs. 12 months DAPT (aspirin + ticagrelor) | All-cause death, MI, definite or probable ST, stroke, or major bleeding (NACE) at 1 year | Abbreviated DAPT achieved non-inferiority and superiority for the primary endpoint (2.8% vs. 5.2%; HR 0.54; 95%CI 0.37–0.80; P for non-inferiority <0.001; P for superiority = 0.002). | Abbreviated DAPT reduced BARC ≥3 bleeding (1.2% vs. 3.4%; HR 0.35; 95%CI 0.20–0.61; P<0.001). No differences between groups in MACE. |
Exclusion of patients with increased bleeding risk Lower than expected event rates. |
| ULTIMATE DAPT (2024) | 3400 | East-Asian (Chinese) 88% | 1 month DAPT (aspirin + ticagrelor) followed by 11 months ticagrelor vs. 12 months DAPT (aspirin + ticagrelor) | Two co-primary endpoints at 12 months: • BARC ≥ 2 bleeding. • MACE: cardiac death, MI, ischaemic stroke, definite ST, or clinically driven target vessel revascularization | Abbreviated DAPT significantly reduced BARC ≥2 bleeding (2.1% vs. 4.6%; HR 0.45; 95% CI 0.30–0.66; P < 0.001). Abbreviated DAPT achieved non-inferiority for MACE (3.6% vs. 3.7%; HR 0.98; 95%CI 0.69–1.39; P for non-inferiority <0.0001; P for superiority = 0.89). | Significant differences in major bleeding were also observed with other bleeding definitions. Potential interaction of age in MACE subgroup analysis (lower efficacy of SAPT in patients ≥65 years old) |
Patients randomized if event-free at 1 month after PCI Lower than expected rates of ischaemic events |
| HOST-BR (2025) | 4897 | East-Asian (Korean) 100% | HBR stratum: 1 month DAPT (aspirin + P2Y12) followed by SAPT vs. 3 months DAPT (aspirin + P2Y12) followed by SAPT Non-HBR stratum: 3 months DAPT (aspirin + P2Y12) followed by SAPT vs. 12 months DAPT (aspirin + P2Y12) |
Three ranked co-primary endpoints at 1 year (tested in hierarchical order): • NACE: all-cause death, MI, ST, stroke, or major bleeding • MACCE: CV death, MI, definite or probable ST, or ischaemic stroke • Any actionable non-surgical bleeding | In the HBR stratum, 1-month DAPT did not reach non-inferiority for NACE (18.4% vs. 14.0%; HR 1.337; 95%CI 1.043–1.713; P for non-inferiority = 0.82) compared to 3-month DAPT. Statistical testing was not formally done for MACCE (9.8% vs. 5.8%; HR 1.724; 95%CI 1.187–2.504) and bleeding (13.8% vs. 15.8%; HR 0.853, 95%CI 0.657–1.107) as non-inferiority of the first primary endpoint was not met. In the non-HBR stratum, 3-months DAPT achieved non-inferiority for NACE (2.9% vs. 4.4%; HR 0.657; 95%CI 0.455–0.949; P for non-inferiority <0.0001) and MACCE (2.2% vs. 2.3%; HR 0.984; 95%CI 0.622–1.558; P for non-inferiority <0.0082), and superiority for bleeding (7.4% vs. 11.7%; HR 0.631; 95% CI 0.502–0.793; P for superiority <0.0001) when compared to 12-month DAPT. |
Increased rates of ischaemic stroke with 1-month DAPT (3.1% vs. 1.1%; HR 2.824; 95%CI 1.318–6.050) in the HBR stratum. Significant differences in bleeding were also observed with other bleeding definitions in the non-HBR stratum. |
SAPT in abbreviated DAPT arms with P2Y12 inhibitor in ~75–80% of subjects (clopidogrel mainly) Ischaemic event rates much higher in the HBR stratum Higher than expected event rates in the HBR stratum and lower than expected event rates in the non-HBR stratum |
| 4D-ACS trial (2025) | 656 | East-Asian (Korean) 100% | 1 month DAPT (aspirin + prasugrel 10 mg) followed by prasugrel 5 mg until 1 year vs. 1 month DAPT (aspirin + prasugrel 10 mg) followed by 11 months DAPT (aspirin + prasugrel 5 mg) | All-cause death, non-fatal MI, stroke, ischaemia-driven target vessel revascularization, and BARC type 2–5 bleeding | Abbreviated DAPT achieved non-inferiority and superiority for the primary endpoint (4.9% vs. 8.8%; HR 0.51; 95%CI 0.27–0.95; P for non-inferiority 0.014; P for superiority = 0.034). | Abbreviated DAPT reduced any bleeding (1.2% vs. 5.2%; HR 0.23; 95%CI 0.08–0.69; P = 0.009) and major bleeding (0.6% vs. 4.6%; HR 0.13; 95%CI 0.03–0.58; P=0.007). No differences between groups in ischaemic outcomes |
Small sample size (adjustment after interim analysis) Reduced dose of prasugrel in both arms |
| NEO-MINDSET trial (2025) | 3410 | South-America (Brazil) 100% White 68.5% Black 18.8% Mixed race 12.7% | 12 months SAPT (potent P2Y12 inhibitor) vs. 12 months DAPT (aspirin + potent P2Y12 inhibitor) | Two ranked primary endpoints at 1 year (tested in hierarchical order): • All-cause death, MI, stroke, or urgent target vessel revascularization • Major or clinically relevant non-major bleeding (BARC type 2, 3, or 5) | SAPT with a potent P2Y12 inhibitor did not achieve non-inferiority (7.0% vs. 5.5%; HR 1.28; 95%CI 0.98–1.68; P for non-inferiority = 0.11). Statistical testing was not formally performed for major or clinically relevant non-major bleeding (2.0% vs. 4.9%; HR 0.40; 95%CI 0.26–0.59) as non-inferiority of the first primary endpoint was not met. | Numerically higher rates of ST in the SAPT arm (0.7% vs. 0.2%; HR 2.99; 95%CI 0.97–9.28) | Patients undergoing successful PCI were randomized within the first 4 of hospitalization. Approximately 70% use of prasugrel. Lower than expected event rates. |
| TARGET-FIRST trial (2025) | 1942 | European 100% | 1 month DAPT (aspirin + P2Y12 inhibitor) followed by 11 months P2Y12 monotherapy vs. 12 months DAPT (aspirin + P2Y12 inhibitor) | All-cause death, MI, ST, stroke, or major bleeding (BARC type 3 or 5) | Abbreviated DAPT achieved non-inferiority and superiority for the primary endpoint (2.1% vs. 2.2%; absolute risk difference 0.09; 95%CI −1.39 to 1.20; P for non-inferiority = 0.02). | Abbreviated DAPT reduced BARC type 2, 3, or 5 bleeding (2.6% vs. 5.6%; HR 0.46; 95% CI 0.29–0.75; P=0.002). Non-inferiority for the primary endpoint was shown in the intention-to-treat and as-treated analyses, but not in the per-protocol analysis. |
Patients randomized if complete revascularization within 7 days and event-free at 1 month after index event. Approximately 74% use of ticagrelor. Very low rates of ischaemic events (lower than expected). |
| ACS, acute coronary syndrome; BARC, bleeding academic research consortium; CI, confidence interval; CV, cardiovascular; DAPT, dual antiplatelet therapy; GUSTO, global use of strategies to open occluded coronary arteries; HBR, high bleeding risk; HPR, high on-treatment platelet reactivity; HR, hazard ratio; LPR, low on-treatment platelet reactivity; MACCE, major adverse cardiac or cerebral events; MACE, major adverse cardiovascular events; MI, myocardial infarction; NACE, net adverse clinical events; PCI, percutaneous coronary intervention; SAPT, single antiplatelet therapy ST, stent thrombosis; STEMI, ST-elevation myocardial infarction; TIMI, thrombolysis in myocardial infarction. | |||||||
Personalized dual antiplatelet therapy abbreviation followed by P2Y12 inhibitor based on bleeding risk
The HOST-BR study evaluated the optimal DAPT duration in patients post-PCI (57% post-ACS) stratified by bleeding risk based on the ARC criteria.77 Patients in the HBR group were randomized to 1 or 3 months of DAPT, and those in the non-HBR stratum were randomized to 3 or 12 months of DAPT. In HBR patients, 1-month DAPT was not non-inferior to 3-month DAPT for NACE, with an excess of ischaemic events in the 1-month DAPT group. In patients without HBR, 3-month DAPT was non-inferior to 12-month DAPT for NACE and MACCE, and superior for bleeding.77
Abbreviated dual antiplatelet therapy followed by monotherapy with aspirin vs. P2Y12 inhibitor
There are no head-to-head trials comparing the strategies of abbreviated DAPT followed by aspirin vs. that followed by P2Y12 inhibitor monotherapy. In a recent large-scale network meta-analysis encompassing 139 086 patients, in comparison to DAPT with aspirin and clopidogrel, aspirin monotherapy significantly reduced bleeding at 12 months [HR 0.71, 95% CI (0.53-0.96)].83 Monotherapy with clopidogrel or ticagrelor did not significantly increase bleeding compared to aspirin. Compared to monotherapy with aspirin, a strategy of abbreviated DAPT followed by P2Y12 inhibitor monotherapy, especially ticagrelor, was associated with a lower risk of myocardial infarction within and after 12 months. Within 12 months, compared to monotherapy with aspirin, monotherapy with ticagrelor was associated with a significant reduction in myocardial infarction [HR 0.69, 95% CI (0.50-0.95)] without increasing bleeding.83 Conversely, in a subanalysis of the GLOBAL LEADERS trial, ticagrelor monotherapy was associated with a reduced risk of ischaemic events (driven by a reduction in myocardial infarction) compared to aspirin monotherapy in the second year post-PCI, at the cost of increased bleeding (BARC 2, 3, or 5).84
De-escalation of dual antiplatelet therapy intensity
Several trials aimed to improve clinical outcomes by tailoring the type or dose of antiplatelet therapy to the individual patient by modifying DAPT regimens, either guided or unguided by genotyping or PFT.16 The term ‘de-escalation’ of DAPT intensity refers to a down-grading of DAPT intensity, from standard DAPT (comprising aspirin plus ticagrelor 90 mg twice daily or aspirin plus prasugrel 10 mg daily) to a less potent antiplatelet regimen of either aspirin plus clopidogrel 75 mg daily or aspirin plus a reduced dose of potent P2Y12 inhibitor.
De-escalation of dual antiplatelet therapy guided by platelet function tests
In patients undergoing PCI, PFT have been shown to be useful for predicting bleeding, ST, and mortality.8586 PFT allows ex vivo measurement of platelet reactivity either in a laboratory or at the bedside.2087 Trials have therefore explored the potential of intensifying antiplatelet treatment based on the results of PFT, but reported no benefit on clinical outcomes. Compared to standard DAPT, de-escalation of P2Y12 inhibitors may mitigate the risk of bleeding, while preserving ischaemic protection after an ACS. PFT-guided de-escalation theoretically holds the advantage of measuring the specific target for antiplatelet therapy but, unlike genotyping, PFT is hampered by variability in platelet function.8788 This creates a challenge in correctly classifying low and normal responders to antiplatelet therapy and, therefore, the ability of PFT to guide long-term DAPT. Two studies specifically explored PFT-guided DAPT de-escalation in patients with ACS (Table 4).8990 In the ANTARCTIC trial, use of PFT to adjust P2Y12 inhibitor treatment in elderly patients with ACS treated with PCI did not improve the net clinical outcome, comprising a composite of major ischaemic and bleeding risks.90 The TROPICAL-ACS trial showed that PFT-guided de-escalation of antiplatelet treatment was non-inferior to standard treatment with prasugrel at 1 year after PCI in terms of net clinical benefit.89
Table 4. Main randomized clinical trials evaluating a strategy of de-escalation of DAPT intensity in patients with ACS
| Study (year) | n | Ethnicity | Treatment groups | Primary endpoint | Main result | Other relevant efficacy or safety endpoints | Comments |
|---|---|---|---|---|---|---|---|
| Unguided de-escalation of DAPT intensity | |||||||
| TOPIC (2017) | 646 | Western (European) 100% | Aspirin + potent P2Y12 inhibitor (ticagrelor or prasugrel) for 1 month followed by aspirin + clopidogrel for 11 months vs. Aspirin + potent P2Y12 inhibitor (ticagrelor or prasugrel) for 12 months | CV death, urgent revascularization, stroke, and BARC ≥2 bleeding at 1 year. | De-escalation strategy diminished the rate of the primary endpoint (13.4% vs. 26.3%; HR 0.48; 95% CI 0.34–0.68; P<0.01), driven by a reduction in bleeding events. | No significant differences in ischaemic outcomes (9.3% vs. 11.5%; P=0.36) Reduction in BARC ≥2 bleeding (4.0% vs. 14.9%; HR 0.30; 95%CI 0.18–0.50; P<0.01) | Single-centre study Small sample size (limited statistical power) |
| HOST-REDUCE-POLYTECH-ACS (2020) | 2338 | East-Asian (South Korea) 100% | Aspirin + prasugrel 10 mg for 1 month followed by aspirin + prasugrel 5 mg for 11 months vs. Aspirin + prasugrel 10 mg for 12 months | All-cause death, non-fatal MI, ST, repeat revascularization, stroke, and BARC ≥2 bleeding at 1 year | De-escalation strategy reduced the rate of the primary endpoint (7.2% vs. 10.1%; HR 0.70; 95% CI 0.52–0.92], P for non-inferiority <0.0001; P for equivalence = 0.012), driven by a reduction in bleeding events. | No increase in ischaemic risk in the de-escalation group compared with the conventional group (HR 0.76; 95% CI 0.40–1.45; P=0·40) Risk of BARC ≥2 bleeding events was significantly decreased with the de-escalation strategy [2.9% vs. 5.9%; 0.48 (0.32–0.73); P=0.0007] |
Unblinded Incidence of the primary endpoint was lower than estimated |
| TALOS-AMI (2021) | 2697 | East-Asian (South Korea) 100% | Aspirin + ticagrelor 90 mg bid followed by aspirin + clopidogrel vs. Aspirin + ticagrelor 90 mg big for 12 months | CV death, MI, stroke, or BARC ≥2 bleeding from 1 to 12 months | Unguided de-escalation diminished the rate of the primary endpoint (4.6% vs. 8.2%; HR 0.55; 95%CI 0.40–0.76; P for non-inferiorityP | =0.15). BARC ≥2 bleeding occurred less frequently in the de-escalation group (3.0% vs. 5.6%, HR 0.52; 95%CI 0.35–0.77, P=0.0012). | Unblinded Randomization after 1 month treatment with aspirin and ticagrelor without major ischaemic or bleeding events Conducted only in East-Asian population (South Korea) |
| Guided de-escalation of DAPT intensity | |||||||
| ANTARCTIC (2016) | 877 | Western (European), % not available | Aspirin (12 months) + prasugrel 5 mg for 2 weeks followed by PFT-guided therapy (prasugrel 10 mg if HPR, clopidogrel 75 mg if LPR, and prasugrel 5 mg if on therapeutic window) for 2 weeks followed by a second PFT-guided adjustment (prasugrel 5 mg if LPR with prasugrel 10 mg, or HPR with clopidogrel 75 mg; continuing therapy for other situations) for 11 months vs. Aspirin plus prasugrel 5 mg for 12 months | CV death, MI, stroke, stent thrombosis, urgent revascularization, and BARC ≥ 2 bleeding at 1 year | No reduction with the PFT-guided strategy of the primary composite endpoint between groups (28% vs. 28%; HR: 1.00, 95%CI 0.78–1.29; P=0.98) | No significant differences in ischaemic endpoints, such as in a key composite secondary endpoint of CV death, MI, ST, or urgent revascularization (10% vs. 9%; HR 1.06; 95%CI 0.69–1.62; P=0.80) No differences in bleeding outcomes, such as in any bleeding (38% vs. 39%; HR 0.98; 95%CI 0.79–1.22; P=0.87) |
Patients aged ≥75 years Small sample size and incomplete monitoring in several patients Platelet function testing (VerifyNow) led to a change of treatment in 45% of patients who were identified as overtreated or undertreated |
| TROPICAL-ACS (2017) | 2610 | Western (European), White 99% | Aspirin for 12 months + prasugrel 10 mg od (or 5 mg according to age and weight) for 1 week followed by clopidogrel 75 mg for 1 week and PFT-guided maintenance therapy (continuing clopidogrel or escalating to prasugrel if HPR) for 11.5 months vs. Aspirin + prasugrel 10 mg (or 5 mg according to age and weight) od for 12 months | CV death, MI, stroke, and BARC ≥ 2 bleeding at 1 year | PFT-guided de-escalation strategy achieved non-inferiority for the primary endpoint (7% vs. 9%; HR 0.81; 95%CI 0.62–1.06; P for non-inferiority = 0.0004, P for superiority = 0.12). | No significant differences in ischaemic outcomes (3% vs.3%; HR 0.77; 95%CI 0.48–1.21; P for non-inferiority = 0.0115) No significant differences in BARC ≥2 bleeding (5% vs. 6%; HR 0.82; 95%CI 0.59–1.13; P=0.23) |
Wide non-inferiority margin Re-escalation (if HPR to clopidogrel) might affect the risk of bleeding |
| POPular Genetics (2019) | 2488 | Western (European), % not available | Aspirin + genotype-guided selection of P2Y12 inhibitor (prasugrel or ticagrelor if carriers of a CYP2C19 loss-of-function allele, clopidogrel in non-carriers) for 12 months vs. aspirin + ticagrelor or prasugrel for 12 months | Two primary outcomes: NACE (all-cause death, MI, definite ST, stroke, or PLATO major bleeding at 1 year) PLATO major or minor bleeding at 1 year | The genotype-guided selection strategy achieved non-inferiority for NACE (5.1% vs. 5.9%; HR 0.87, 95%CI 0.62– 1.21; P for non-inferiority <0.001; P=0.40 for superiority). Reduction in the primary safety endpoint for bleeding with the genotype-guided regimen (9.8% vs. 12.5%; HR 0.78, 95%CI 0.61–0.98; P=0.04) | No significant differences in ischaemic endpoints between groups | Wide non-inferiority margin due to a lower than anticipated incidence of the combined primary endpoint. Treatment until genotyping (up to 48 h after primary PCI) at discretion of the treating physician. |
| ACS, acute coronary syndrome; BARC, bleeding academic research consortium; CI, confidence interval; CV, cardiovascular; HPR, high on-treatment platelet reactivity; HR, hazard ratio; LPR, low on-treatment platelet reactivity; MI, myocardial infarction; NACE, net adverse clinical events; ST, stent thrombosis. | |||||||
De-escalation of dual antiplatelet therapy guided by genotyping
Clopidogrel is a prodrug, metabolized into its active form in the liver, mainly by a cytochrome P450 (CYP) enzyme, the effectiveness of which is determined by the CYP2C19 gene. The CYP2C19 gene has polymorphism, and its loss-of-function alleles are associated with a reduced metabolism of clopidogrel, resulting in increased cardiovascular ischaemic risk. The Popular Genetics trial compared a genotype-guided P2Y12-inhibition de-escalation strategy with standard DAPT consisting of aspirin plus ticagrelor/ prasugrel (Table 4). The study included patients with STEMI undergoing primary PCI, with genotyping performed during the acute admission. In the genotype-guided group, CYP2C19 loss-of-function allele carriers received ticagrelor (43.1%) or prasugrel (1.2%) and non-carriers received clopidogrel (55.5%) as part of DAPT with aspirin. Genotype-guided clopidogrel was superior to standard care with ticagrelor (90.2%) or prasugrel (2.2%) in relation to PLATO major or minor bleeding. The composite of all-cause death, myocardial infarction, ST, stroke, and PLATO major bleeding was numerically lower in the genotype-guided group compared to the standard treatment group, although not reaching statistical significance.91
Unguided de-escalation of dual antiplatelet therapy
Unguided de-escalation was compared against standard DAPT in three RCTs after ACS (Table 4).929394 In the TOPIC trial, ACS patients were randomized to clopidogrel-based DAPT vs. standard DAPT after a period of 1-month DAPT with either ticagrelor or prasugrel.92 The primary composite endpoint of cardiovascular death, urgent revascularization, stroke, and BARC bleeding grade ≥2 at 1-year post-ACS was significantly lower in the de-escalation arm compared to the standard DAPT arm, mainly due to a lower rate of BARC ≥2 bleeding, while ischaemic events were similar in both arms.92 The HOST-REDUCE-POLYTECH-ACS trial tested the non-inferiority of reducing the dose of prasugrel (from 10 to 5 mg) 1 month after ACS in East Asian patients.93 The primary endpoint of net adverse clinical events (all-cause death, non-fatal myocardial infarction, ST, repeat revascularization, stroke, and BARC ≥2 bleeding) at 1 year was similar in the two arms, meeting the criteria for non-inferiority. Ischaemic events were not increased, and bleeding events were significantly lower in the de-escalation arm,93 irrespective of PCI complexity.95 In TALOS-AMI, an open-label, non-inferiority trial, 2697 East-Asian patients were randomized 1 month after ACS to clopidogrel-based DAPT or continuation of ticagrelor-based DAPT. The de-escalation strategy met the criteria for non-inferiority and superiority for the primary composite endpoint of CV death, myocardial infarction, stroke, or BARC ≥2 bleeding, with reduced BARC ≥2 bleeding in the de-escalation group.94 In a recent network meta-analysis, the magnitude of reduction in bleeding appeared greater with unguided compared to the guided de-escalation.96
Other measures to reduce bleeding
In addition to use of patient-tailored DAPT, it is also crucial to implement more general measures to mitigate bleeding risk and prevent unnecessary early interruption of DAPT, a significant driver of mortality.97 Arterial access should routinely be performed radially,1 non-urgent (invasive) procedures should be deferred whenever possible, and blood pressure should be optimally controlled.98 Concurrent platelet inhibitors (chronic non-steroidal anti-inflammatory drugs, selective serotonin re-uptake inhibitors) should be avoided as much as possible, and non-steroidal agents should be switched to simple analgesics such as paracetamol or anti-inflammatories such as celecoxib and meloxicam if required.99 Proton pump inhibitors are advised for patients on DAPT at higher-than-average risk of gastrointestinal bleeding (e.g. history of gastrointestinal ulcer/haemorrhage, OAC, chronic non-steroidal anti-inflammatory drug/ corticosteroid use), or with two or more of the following: (i) age ≥65 years, (ii) dyspepsia, (iii) gastro-oesophageal reflux disease, (iv) Helicobacter pylori infection, or (v) chronic alcohol use.1 Omeprazole and esomeprazole inhibit CYP2C19 and might reduce the effect of clopidogrel; therefore, coadministration (certainly in case of clopidogrel monotherapy) should be avoided.100101 Additionally, consideration should be given to the patient profile, since a frail or critically ill patient (sedated, enterally fed, shock status) may require a different approach compared to a lower-risk individual.102
GT 04

GT 04.
Therapeutic options to minimize ischaemic risk
Options to reduce ischaemic events in very high-risk individuals are shown in Figure 3.
Very low dose rivaroxaban in the first year post-acute coronary syndrome
The rationale behind the addition of very low dose rivaroxaban (VLDR) 2.5 mg twice daily to DAPT, comprising aspirin and clopidogrel, in the setting of ACS stems from the observation that rivaroxaban reduces thrombin generation,103104 which may further reduce ischaemic events,105 although it does not improve the fibrinolytic profile.106 In the Rivaroxaban in Patients with a Recent Acute Coronary Syndrome (ATLAS ACS 2–TIMI 51) trial in 15 000 patients, the addition of VLDR (2.5 or 5 mg twice daily) to DAPT reduced the risk of cardiovascular death compared to placebo, at the expense of a four-fold increase in major bleeding.107 Comparable risk estimates were observed in the STEMI subgroup.108 As ischaemic risk predominates over bleeding early post-ACS, there may be a short window to use VLDR to reduce ischaemic or atherothrombotic events, without increasing bleeding. A different approach was evaluated in one study, which suggested VLDR may be a substitute for aspirin in stabilized ACS patients after PCI.109
Prolonged dual antithrombotic therapy options beyond 1 year
Large RCTs have established the benefits of long-term intensified antithrombotic therapy in the prevention of MACE in patients with a history of myocardial infarction or other atherothrombotic conditions, whilst consistently showing increase in bleeding (Table 5). The CHARISMA trial included patients with either increased risk of cardiovascular disease or a history of symptomatic atherosclerotic disease, and showed that the addition of clopidogrel to background aspirin therapy did not significantly reduce MACE, but showed a signal for benefit in those with prior myocardial infarction, stroke, or symptomatic peripheral arterial disease.110111 The DAPT trial showed that prolonging thienopyridine therapy beyond 12 months after PCI was associated with reduced risks of either ST or MACE at 30 months,112 although this trial was conducted in an era of higher rates of ST compared with contemporary practice. Prolonged thienopyridine therapy was associated with a numerically higher rate of all-cause death. The PEGASUS-TIMI 54 trial was the only large placebo-controlled RCT to focus purely on a population with a history of myocardial infarction more than 12 months prior and assessed the effects of ticagrelor 90 mg or 60 mg twice-daily compared to placebo, on a background of aspirin therapy.113 Both doses of ticagrelor achieved similar reduction in MACE and similar increase in bleeding complications, without any significant impact on all-cause mortality. The benefit of ticagrelor seemed to be confined to those who did not have HBR, and there was evidence of cardiovascular mortality reduction in those with high ischaemic risk without HBR.114 The COMPASS trial included patients with stable atherosclerotic vascular disease and compared two rivaroxaban-based regimens, namely dual antithrombotic therapy with rivaroxaban (2.5 mg twice daily) plus aspirin and rivaroxaban monotherapy (5 mg twice daily), with aspirin monotherapy.115 Dual therapy with aspirin plus rivaroxaban significantly reduced MACE at the cost of a similar increase in major bleeding, whereas the rivaroxaban-alone regimen did not improve cardiovascular outcomes compared to aspirin and resulted in more major bleeding events.115
GT 05
Table 5. Main randomized clinical trials evaluating a strategy of prolonged dual antithrombotic therapy.
| Study (year) | n | Ethnicity | Treatment groups | Primary endpoint | Main result | Other relevant efficacy or safety endpoints | Comments |
|---|---|---|---|---|---|---|---|
| CHARISMA (2006) | 15 603 | White 80% Hispanic 10% Asian 5% Black 3% | DAPT with aspirin + clopidogrel vs. SAPT with aspirin | CV death, MI, or stroke at median 28 months | The DAPT strategy did not significantly reduce primary endpoint (6.8% vs. 7.3%; RR 0.93; 95%CI 0.83–1.05; P=0.22). | DAPT did not significantly increase severe bleeding (1.7% vs. 1.3%; P=0.09) or fatal bleeding (0.3% vs. 0.2%; P=0.17) but increased the rate of moderate bleeding (2.1% vs. 1.3%; RR 1.62, 95%CI 1.27–2.08; P<0.001). Benefit of DAPT in the subgroup of patients (n = 9478) with prior MI, stroke, or symptomatic PAD (7.3% vs. 8.8%; HR 0.83; 95% CI 0.72– 0.96; P=0.01). |
Patients with established or high risk of CV disease (3846 prior MI) Lack of efficacy in patients without a history of atherothrombotic event. |
| DAPT (2014) | 9961 | White 91% | DAPT with aspirin and either clopidogrel or prasugrel vs. SAPT with aspirin | Coprimary endpoints at 30 months after PCI: (1) Definite or probable ST (2) Death, MI, or stroke | Prolonged DAPT reduced the rates of ST (0.4% vs. 1.4%; HR 0.29, 95%CI 0.17–0.48; P< 0.001) and the composite of death, MI, or stroke (4.3% vs. 5.9%; HR 0.71; 95%CI 0.59–0.85; P<0.001). | The rate of MI was lower with DAPT (2.1% vs. 4.1%; HR 0.47; P<0.001). The rate of all-cause death was 2.0% with DAPT vs. 1.5% with SAPT (HR 1.36; 95% CI 1.00–1.85; P=0.05). Moderate or severe bleeding was increased with DAPT (2.5% vs. 1.6%, P=0.001). In the subgroup of patients presenting with MI (n=3576), prolonged DAPT reduced rates of definite or probable ST (0.5% vs. 1.9%; HR 0.27, 95%CI 0.13–0.57; P < 0.001) and MACCE (3.9% vs. 6.8%; HR 0.56, 95%CI 0.42–0.76; P<0.001). |
Patients randomized 12 months after PCI. Older stent designs associated with higher rates of ST. |
| PEGASUS-TIMI 54 (2015) | 21 162 | White 86% | DAPT with aspirin + ticagrelor 90 mg bid vs. DAPT with aspirin + ticagrelor 60 mg bid vs. SAPT with aspirin | CV death, MI, or stroke at 3 years | The prolonged DAPT strategy with both ticagrelor doses reduced the combined ischaemic primary endpoint compared to placebo: ticagrelor 90 mg bid (7.85% vs. 9.04%; HR 0.85; 95%CI 0.75–0.96; P=0.008) and 60 mg bid (7.77% vs. 9.04%; HR 0.84; 95%CI 0.74–0.95; P=0.004). | Prolonged DAPT increased the rates of TIMI major bleeding compared to placebo: ticagrelor 90 mg (2.60% vs. 1.06%; HR 2.69; 95%CI 1.96– 3.70; P<0.001) and 60 mg (2.30% vs. 1.06%; HR 2.32; 95%CI 1.68– 3.21; P<0.001). |
Patients enrolled 1–3 years after suffering an MI. Better safety profile with the 60 mg bid dose. Benefit seen only in those without HBR. |
| COMPASS (2017) | 27 395 | White 62% Black 1% Asian 16% Other 21% | DAT with aspirin + rivaroxaban 2.5 mg bid vs. rivaroxaban 5 mg bid vs., aspirin | CV death, MI, or stroke at mean follow-up of 23 months | DAT with aspirin + rivaroxaban reduced the rate of the primary endpoint compared to aspirin alone (4.1% vs. 5.4%; HR 0.76, 95%CI 0.66–0.86; P<0.001). | DAT increased the rates of major bleeding when compared to aspirin alone (3.1% vs. 1.9%; HR 1.70; 95% CI 1.40–2.05; P<0.001). The rivaroxaban-alone treatment did not reduced cardiovascular ischaemic outcomes compared to aspirin-alone therapy, but augmented major bleeding events | Patients with stable atherosclerotic vascular disease. Stopped early due to efficacy, which may overestimate the treatment effect. |
| CI, confidence interval; CV, cardiovascular; DAPT, dual antiplatelet therapy; DAT, dual antithrombotic therapy; HR, hazard ratio; MI, myocardial infarction; MACCE, major adverse cardiovascular and cerebrovascular endpoints; NACE, net adverse clinical events; PAD, peripheral artery disease; RR, relative risk; ST, stent thrombosis; TIMI, thrombolysis in myocardial infarction. | |||||||

GT 05.
Specific advice according to…
Ethnicity
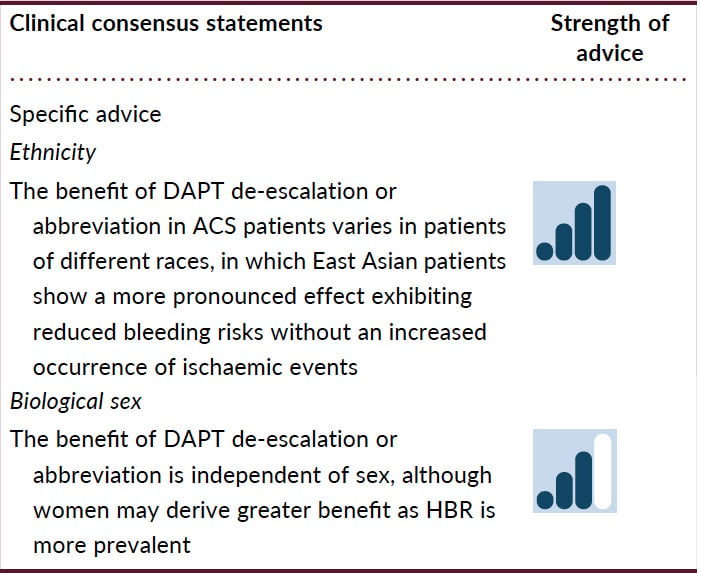
Inherited abnormalities in the coagulation-fibrinolysis axis and inflammation play crucial roles in the progression of atherosclerotic cardiovascular disease, with African Americans experiencing the highest level of atherothrombotic risk and East Asians the lowest (Table 6).116 A pooled analysis of 10 RCTs involving 22 638 patients undergoing PCI evaluated racial differences in ischaemic endpoints (death, myocardial infarction, and ischaemia-driven revascularization).117 Five-year major CV event rates were 18.8% in white patients (reference group), 23.9% in black, 21.5% in Hispanic, and 11.2% in Asian patients. Multivariate analysis demonstrated that black race was an independent predictor of cardiovascular events.
The distinct balance between reduced ischaemic benefit and increased bleeding risk between patients of different races may influence the optimal DAPT strategy.118 In a meta-analysis including 23 trials, ‘de-escalation DAPT strategy’ showed differential clinical benefits between East Asian and Caucasian patients compared with conventional DAPT strategy.119 Unguided de-escalation of DAPT intensity has been shown to reduce bleeding without an increase in ischaemic events; however, most of these studies were conducted in East Asian patients and in only 2 small studies in non-East Asian patients.16 While a reduction of DAPT intensity can minimize bleeding without safety concerns in East Asian patients, in Caucasian patients, reduction of DAPT intensity could incur an ischaemic penalty. Abbreviating DAPT duration has been shown to reduce net adverse clinical events driven by a reduction in major bleeding. However, whilst this approach appears equally safe in terms of not increasing ischemic events in both East Asian and non-East Asian patients, the reduction in major bleeding with this strategy was mainly seen in East Asian patients, where abbreviated DAPT strategy substantially reduced major bleeding without increasing MACE.
Table 6. Racial differences in different factors associated with thrombogenicity.
| Factors | Racial difference | Mechanism |
|---|---|---|
| Lipid profile | ||
| High LDL-cholesterol | Hispanics = Caucasians > East Asians > African Americans | Atherogenesis |
| Low HDL-cholesterol | Hispanics > Caucasians ≥ East Asians > African Americans | Atherogenesis |
| High triglyceride | East Asians > Hispanics > Caucasians > African Americans | Atherogenesis |
| Lipoprotein(a) level | African Americans > Caucasians > Hispanics > East Asians | Atherogenesis |
| Inflammation | ||
| CRP level | African Americans > Hispanics > Caucasians > East Asians | Inflammation |
| Platelets | ||
| Platelet count | African Americans > Hispanics > Caucasians | Haemostasis |
| Regulation of Gq pathway after PAR-4 stimulation | African Americans > Caucasians | Platelet activation |
| Responsiveness to clopidogrel | Caucasians > African Americans > East Asians | Antiplatelet efficacy |
| Responsiveness to ticagrelor | East Asians > Caucasians > African Americans | Antiplatelet efficacy |
| Coagulation | ||
| Factor V Leiden mutation | Caucasians (~5%) > African Americans (~1.5%) > Hispanics = Asians | Coagulant activity |
| Prothrombin G20210A mutation | Caucasians (2–5%) > African Americans = Hispanics = Asians | Coagulant activity |
| von Willebrand factor level | African Americans > Caucasians = Hispanics ≥ East Asians | Endothelial adhesion factor |
| Fibrinogen, factor VII and VIII, and thrombin levels | African Americans ≥ Caucasians = Hispanics ≥ East Asians | Coagulant activity |
| D-dimer level | African Americans > Caucasians = Hispanics > East Asians | Prothrombotic and hypofibrinolytic state |
| Anticlotting activity | ||
| Effect of activated protein C | Asians = Hispanics ≥ African Americans > Caucasians | Anti-coagulant activity |
| PAI-1 level | Caucasians > Hispanics > East Asians ≥ African Americans | Anti-fibrinolytic activity |
| APC, activated protein C; CRP, C-reactive protein; HDL, high-density lipoprotein; LDL, low-density lipoprotein; PAI −1, plasminogen activator inhibitor-1: PAR, proteinase-activated receptor. | ||
Biological sex
There are well-characterized sex-related differences in ischaemic and bleeding risks. Amongst patients with ACS, women tend to be 5–7 years older than men, with more comorbidities.120 Frequent late ACS diagnosis in women can contribute to the delay in the initiation of antiplatelet treatment, which may contribute to worse outcomes. Evidence supports similar efficacy of antithrombotic drugs in women and men, and the trials of de-escalation or abbreviation, although including more men, did not show any sex-related differences in outcomes. Importantly, studies have shown that female patients may receive higher antithrombotic drug dosing than appropriate for their kidney function and body weight, both of which translate into an increase in bleeding rates.121 Additionally, femoral arterial access was used up to 50% more in women than in men, and corresponded with excessive bleeding.122123124 Accordingly, the Academic Research Consortium HBR score was significantly higher among women than men (due to older age and more comorbidities in women), which translates into 43% of women and 32% of men being classified as HBR.125
GT 06
GT 06.
Practical advice on individualizing antithrombotic strategy: what to do if…?
Average ischaemic and bleeding risks
The 2023 ESC guidelines for the management of ACS recommend 12 months of DAPT as the default strategy in all patients, unless there is HBR.1 This recommendation is based on the duration of DAPT in the pivotal RCTs on DAPT.563381 The more potent P2Y12 inhibitors prasugrel and ticagrelor are recommended over clopidogrel.
In patients who are event-free after 3–6 months of DAPT and who are not at high ischaemic risk, the ESC guidelines for ACS management suggest that single antiplatelet therapy (preferably with a P2Y12 inhibitor) should be considered.1 However, experts in the field have argued against the default strategy of the ESC guidelines and advocate a shorter period of 1–3 months of DAPT as the default strategy, followed by single antiplatelet therapy with ticagrelor,126127 based on the results of RCTs and individual patient level meta-analyses showing such a strategy to reduce bleeding without increasing ischaemic events.81127128
Ischaemic risk exceeds bleeding risk
The 2023 ESC guidelines recommend 12 months of DAPT duration in patients with high ischaemic risk and low bleeding risk. Furthermore, continuation of a second antithrombotic agent, in addition to aspirin, for extended long-term secondary prevention should be considered in these patients.1 For patients at increased risk of ischaemic events and low bleeding risk, continuing long-term DAPT after 1 year may be appropriate, with aspirin and ticagrelor 60 mg twice daily being a reasonable option to balance ischaemic and bleeding risks.126 In selected high-risk individuals (e.g. prior ischaemic stroke or polyvascular disease), extended dual antithrombotic therapy (aspirin + VLDR) may also be appropriate for reducing ischaemic complications.115
BLEEDING RISK EXCEEDS ISCHAEMIC RISK
Abbreviation and/or de-escalation of the initial DAPT is advisable in patients at HBR. Abbreviating DAPT and continuing single antiplatelet therapy with aspirin (after 3–6 months) or P2Y12-inhibitor (clopidogrel or ticagrelor) (after 1–3 months) has been shown to be effective in reducing bleeding with no obvious increase in ischaemic events. De-escalation of DAPT by switching the more potent P2Y12-inhibitors ticagrelor or prasugrel to clopidogrel, either routinely (after 1–3 months) or guided by the results of genetic tests (after 1–4 weeks), has also been shown to decrease bleeding with no increase in ischaemic events.216126 Beyond 12 months, prolonged DAPT or the combination of aspirin and VLDR should be avoided owing to the associated increase in bleeding compared to aspirin monotherapy.1
High ischaemic and high bleeding risk
High ischaemic and bleeding risk co-exist depending on the definitions used in 5–10% of the patients, and HBR is present in up to 55% of patients undergoing complex PCI (e.g. left main PCI).129130 A pooled analysis of 14 963 patients from eight RCTs with 3118 undergoing complex PCI showed that patients had a higher risk of ischaemic events, but benefited from long-term DAPT only if HBR features were not present.129 Similarly, the effects of 1-month DAPT relative to 12-month DAPT were consistent regardless of HBR and complex PCI in the STOPDAPT-2 ACS trial.131 P2Y12 inhibitor monotherapy after 1–3 months of DAPT was associated with similar rates of fatal and ischaemic events and a lower risk of major bleeding compared with standard DAPT, irrespective of PCI complexity and therefore could be an option in patients at both high bleeding and high ischaemic risk.132 Of note, in the TWILIGHT study, the absolute risk reduction in major bleeding achieved by ticagrelor monotherapy compared to DAPT was higher in HBR than non-HBR patients with multiple ischaemic and angiographic risk factors.133 In a post hoc analysis of the HOST-REDUCE-POLYTECH-ACS study, prasugrel dose de-escalation was not associated with an increased risk of ischaemic outcomes but was associated with fewer bleeding events at 1 year, irrespective of PCI complexity. Taken together, HBR should be prioritized regardless of PCI complexity and ischaemic risk, to guide decision-making on DAPT duration. There are no RCTs comparing different strategies (unguided/guided de-escalation/short DAPT) in HBR/HIR patients.A post hoc exploratory analysis of the PEGASUS-TIMI 54 trial showed that long-term ticagrelor therapy appeared unsuitable for those with prior myocardial infarction/multiple ischaemic risk factors and HBR.114 These data, although exploratory, suggest that HBR/HIR patients do not benefit from extended DAPT strategies.
GT 07

GT 07.
Low ischaemic and low bleeding risk
Current guidelines recommend that patients with a low ischaemic and low bleeding risk should receive standard guideline-directed DAPT, namely 12 months of aspirin and either prasugrel or ticagrelor.1 Both prasugrel and ticagrelor increase bleeding compared to clopidogrel, however the risk-benefit ratios are favourable with NNT of 46 and 53, and NNH of 167 for both drugs.56 However, since bleeding risk may outweigh ischaemic risk when both are low, it would be appropriate to consider DAPT abbreviation or de-escalation in these patients.
Conclusions
The duration and choice of DAPT in ACS patients post-PCI should take into account the thrombotic and bleeding risks of the individual and should be personalized (Graphic Abstract). Several factors underpin the need to consider alternative DAPT regimens, over standard DAPT. First, de-escalation or abbreviation affords significant reduction in bleeding, particularly for the 40% of PCI patients classified as HBR. Second, the risk of ST and recurrent myocardial infarction beyond 1–3 months is very low with use of the latest generation of DES. Third, the prevalence of HBR greatly exceeds high ischaemic risk. Further, amongst patients at HBR, standard DAPT, in comparison to abbreviated or de-escalated DAPT, increases the net risk of major adverse events, even in the presence of high ischaemic risk. Conversely, amongst patients at high ischaemic risk, in the absence of HBR, a prolonged dual antithrombotic regimen may be appropriate to reduce the longer-term atherothrombotic risk.
Although the coexistence of increasing numbers of risk factors for bleeding is associated with a stepwise increase in bleeding risk, suitable scores are not available to guide decision-making regarding antithrombotic therapy that would take into account the relative weight of each HBR criterion. Similarly, there is no evidence that use of a clinical risk score to assess ischaemic risk can guide DAPT strategy to improve clinical outcome. Additionally, since there is no ‘formula’ to calculate and balance ischaemic and bleeding risks, and since many risk factors overlap, focus should be on managing the risk most amenable to modification through pharmacotherapy. In this respect, bleeding risk should dominate the decision making for DAPT duration and/or intensity.
Taking into account that a default strategy of DAPT consisting of a potent P2Y12 receptor inhibitor (prasugrel or ticagrelor) in addition to aspirin for 12 months is recommended in the 2023 ESC guidelines for the management of ACS, this expert committee advises that in patients with ACS undergoing PCI, it is beneficial to personalize the duration and intensity of DAPT treatment. Indeed, guidance by the clinical consensus statements (Figure 4) herein is advised to minimize the occurrence of ischaemic and bleeding events and to improve outcomes.
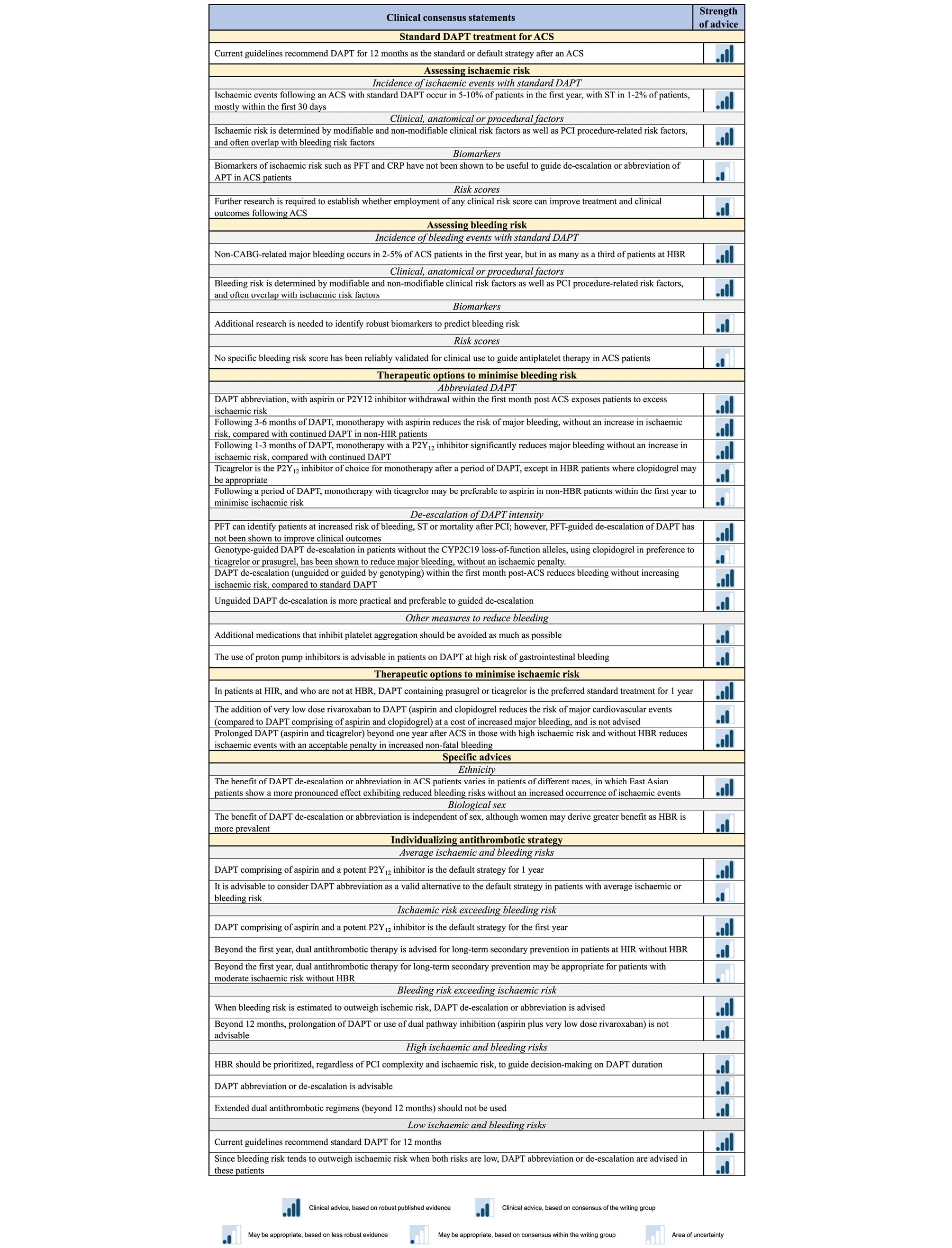
Figure 4. Clinical consensus statements. ACS, acute coronary syndrome; CABG, coronary artery bypass graft; CRP, C-reactive protein; DAPT, dual antiplatelet therapy; HBR, high bleeding risk; HIR, high ischaemic risk; PCI, percutaneous coronary intervention; PFT, platelet function testing.
Author contributions
Jurrien M. ten Berg (Conceptualization, Investigation, Visualization, Writingâ—original draft, Writingâ—review and editing), Gianluca Campo (Conceptualization, Investigation, Visualization, Writingâ—original draft, Writingâ—review and editing), Tobias Geisler (Conceptualization, Investigation, Visualization, Writingâ—original draft, Writingâ—review and editing), Erik L. Grove (Conceptualization, Investigation, Visualization, Writingâ—original draft, Writingâ—review and editing), Sigrun Halvorsen (Conceptualization, Investigation, Visualization, Writingâ—original draft, Writingâ—review and editing), Kurt Huber (Conceptualization, Investigation, Visualization, Writingâ— original draft, Writingâ—review and editing), Young-Hoon Jeong (Conceptualization, Investigation, Visualization, Writingâ— original draft, Writingâ—review and editing), Gregory Y. H. Lip (Conceptualization, Investigation, Visualization, Writingâ— original draft, Writingâ—review and editing), Eliano P. Navarese (Conceptualization, Investigation, Visualization, Writingâ— original draft, Writingâ—review and editing), Andrea Rubboli (Conceptualization, Investigation, Visualization, Writingâ— original draft, Writingâ—review and editing), Jolanta M. Siller Matula (Conceptualization, Investigation, Visualization, Writingâ— original draft, Writingâ—review and editing), Robert F. Storey (Conceptualization, Investigation, Visualization, Writingâ— original draft, Writingâ—review and editing), Marco Valgimigli (Conceptualization, Investigation, Visualization, Writingâ— original draft, Writingâ—review and editing), Christophe Vandenbriele (Conceptualization, Investigation, Visualization, Writingâ— original draft, Writingâ—review and editing), Gemma Vilahur (Conceptualization, Investigation, Visualization, Writingâ— original draft, Writingâ—review and editing), Ingo Ahrens (Conceptualization, Investigation, Visualization, Writingâ— original draft, Writingâ—review and editing), Diana A. Gorog (Conceptualization, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writingâ— original draft, Writingâ—review and editing), and Jose Luis Ferreiro (Conceptualization, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writingâ—original draft, Writingâ—review and editing)
Data availability
There are no new data generated or analyzed in this paper.
Funding
This work was not funded, in whole or in part, for any of the authors.
Conflict of interest statement
D.A.G.: Institutional research grants from Bayer, AstraZeneca, Chiesi. Speaker’s honoraria/advisory board BMS, Janssen, Chiesi, all outside the submitted work. J.t.B.: Institutional research grants from ZonMw (Dutch Government) and Daiichi Sankyo. Speaker’s honoraria/advisory board Daiichi Sankyo, CeleCor, AstraZeneca. G.C.: Institutional research grants from SMT, Siemens Healthcare, GE Healthcare, Abbott Vascular, and GADA outside the present work. T.G.: Speaker/consultancy honoraria from Abbott, AstraZeneca, Boehringer Ingelheim, Boston Scientific, Edwards Lifescience, Ferrer/Chiesi, Haemonetics, Pfizer; institutional research grants from Bayer Healthcare, Bristol Myers Squibb, Daiichi Sankyo, Edwards Lifescience, Ferrer/Chiesi, Medtronic outside of the submitted work. B.G.: None. E.L.G.: Speaker’s honoraria or consultancy fees from AstraZeneca, Bayer, Bristol-Myers Squibb, Pfizer, Novo Nordisk, Lundbeck Pharma, and Organon, and unrestricted research grants from Boehringer Ingelheim. E.L.G. is an investigator in clinical studies sponsored by AstraZeneca, Idorsia, or Bayer. S.H.: Speaker’s honoraria from AstraZeneca, Bayer, Pfizer, and Bristol-Myers Squibb are not related to this work. K.H.: Lecture fees/honoraria from AstraZeneca, Bayer, Bristol-Myers Squibb, Boehringer-Ingelheim, Chiesi, Daiichi Sankyo, Ferrer, and Pfizer. Y.H.J.: Honoraria for lectures from Daiichi Sankyo, Sanofi-Aventis, Han-mi Pharmaceuticals, and Daewoong Pharmaceuticals, and research grants or support from Samjin Pharmaceuticals, Hanmi Pharmaceuticals, Yuhan Pharmaceuticals, Biotronik Korea, and U and I Corporation. G.Y.H.L.: G.Y.H.L. is a National Institute for Health and Care Research (NIHR) Senior Investigator. He is a consultant and speaker for BMS/Pfizer, Boehringer Ingelheim, Daiichi-Sankyo, and Anthos (no fees are received personally). E.P.N.: research grants from Abbott, Amgen, and lecture fees/honoraria from Amgen, AstraZeneca, Bayer, Pfizer, and Sanofi-Regeneron, outside the submitted work. A.R.: None. J.M.S.M.: Speaker or lecture honoraria from Chiesi, OM Pharma, Daiichi Sankyo, Biotronik, Cordis, Terumo. R.F.S.: Institutional research grants/support from AstraZeneca and Cytosorbents; and personal fees from Abbott, Afortiori Development/Thrombolytic Science, Alfasigma, AstraZeneca, Boehringer Ingelheim/Lilly, Bristol Myers Squibb/Johnson & Johnson, Chiesi, Cytosorbents, Daiichi Sankyo, Idorsia, Novartis, Novo Nordisk, Pfizer, PhaseBio, and Tabuk. M.V.: Consultancy and speaker fees from Abbott Vascular, Avimedica, Terumo Inc., Vesalio, Concept Medical, Om Pharma, Johnson & Johnson, Astrazeneca, Sanofi, Daiichi Sankyo, Novartis, Biotronik, Chiesi Pharma, Idorsia; he receives research grants from Concept Medical and Om Pharma. C.V.: Honoraria for lectures from Daiichi Sankyo, Abiomed, and Medtronic. G.V.: Honoraria for lectures for AstraZeneca and unrestricted research grants support from AstraZeneca and Servier. I.A.: Honoraria for lectures from AstraZeneca, Bayer Healthcare, Boehringer Ingelheim, Boston Scientific, Daiichi Sankyo, Eli Lilly Co. J.L.F.: Honoraria for lectures from Eli Lilly Co, Daiichi Sankyo, Inc., AstraZeneca, Pfizer, Abbott, Boehringer Ingelheim, Bristol-Myers Squibb, Rovi, Terumo. and Ferrer; consulting fees from AstraZeneca, Eli Lilly Co., Ferrer, Boston Scientific, Pfizer, Boehringer Ingelheim, Daiichi Sankyo, Inc., Bristol-Myers Squibb, and Biotronik; research grants from AstraZeneca.
