Cory:
Unlock Your AI Assistant Now!
In patients with severe aortic stenosis and a hostile aortic anatomy scheduled for transcatheter aortic valve implantation (TAVI), concerns may arise regarding the risk of vascular complications and a previously reported increased stroke risk12. One practical strategy is to use an extended-length introducer sheath that bypasses the problematic aortic segment(s). However, data supporting this approach remain limited.
In this report, we describe the feasibility, safety, and technical considerations of using a 65 cm long GORE DrySeal Flex introducer sheath (W. L. Gore & Associates) in patients with hostile aortic anatomies undergoing transfemoral (TF)-TAVI with self-expanding valves (SEV). The DrySeal Flex is a fixed-diameter sheath featuring a hydrophilic coating and a long-tapered dilator. Designed for flexibility, the sheath offers sufficient support and kink resistance. Due to its increased length, the sheath is placed over a stiff guidewire and positioned in the left ventricle. The sheath is advanced across the aortic arch and remains in place until delivery of the transcatheter heart valve (THV) at the level of the aortic annulus or until completion of all procedural steps – such as crossing the valve, balloon dilatation, and valve delivery and implantation (Moving image 1).
Among 2,430 patients undergoing TF-TAVI at our institution between 2021 and 2025, the 65 cm DrySeal sheath was utilised in 200 patients (8.2%) with hostile aortic anatomies. Indications for use included severe calcification of the aortic arch (28%), heavy atheromatous or shaggy aorta (20%), acute aortic arch angulation (26%), tortuous aorta (36%), and bicuspid aortic valve with a coarctation-like aorta (8%) (Figure 1). In standard TF-TAVI cases, a 35 cm long introducer sheath or a strategy using an in-line/integrated sheath was utilised. Patients treated with the long DrySeal sheath had a higher prevalence of peripheral arterial disease (20.5% vs 8.7%; p<0.001) and prior stroke (15.5% vs 9.6%; p=0.008), more frequently presented with a bicuspid aortic valve (30.0% vs 11.0%; p<0.001), and had a higher median Society of Thoracic Surgeons risk score (5.4% vs 4.0%; p<0.001), compared with those treated with a 35 cm long sheath or an in-line/integrated sheath, reflecting the higher-risk nature of this patient cohort. The most commonly used sheath size was 18 Fr (45.0%), and the most frequently implanted valve was the Evolut (63.5%; Medtronic). Vascular closure involved a single ProStyle preclosure device combined with an AngioSeal (Terumo) in 178 patients (89.0%), with an additional ProStyle in 18 patients (9.0%) and bailout MANTA (Teleflex) closure in 4 patients (2.0%).
In all cases, the 65 cm DrySeal sheath successfully facilitated delivery and implantation of the THV via TF access. The rates of all-cause mortality, disabling stroke, and major access-related complications were all 0.5%. Notably, there was one fatal ascending aortic dissection, which was reviewed and attributed to multiple Evolut THV repositionings and resheathings in a patient with severely calcific bicuspid aortic stenosis and a horizontal aorta with aortopathy. During one repositioning manoeuvre, the valve jumped towards the ascending aorta while exerting a forward push on the delivery system, thereby creating a dissection in the ascending aorta. When compared with conventional sheath TF-TAVI cases, there were no statistically significant differences in outcomes: all-cause mortality (0.5% vs 0.1%; p=0.23), all stroke (1.5% vs 1.6%; p=0.91), or major vascular complications (1.0% vs 0.8%; p=0.41) (Figure 1).
This single-centre experience demonstrates that a longer introducer sheath can enhance procedural stability and control during TF-TAVI in patients with challenging aortic anatomies. By overcoming significant aortic tortuosity, kinks, or angulations, the extended-length sheath allows for more controlled and precise THV delivery and deployment, thereby not only minimising the risk of direct vascular complications but also enhancing catheter stability and responsiveness. This may reduce the need for valve-repositioning manoeuvres and their associated embolic or vascular complication risks.
Although stroke rates were comparable with our standard TF-TAVI population, it is noteworthy that patients treated with the extended-length sheath constituted a higher-risk cohort. This apparent benefit might be related to the sheath’s ability to shield the vulnerable aortic arch, thus reducing repeated contact of wires, catheters, and devices with calcific atheromatous plaques and potentially limiting embolic release12. However, this observation remains a hypothesis that needs validation in future prospective, ideally randomised, studies.
In conclusion, the use of an extended-length sheath when performing TF-TAVI in patients with a hostile aortic anatomy is feasible and may enhance procedural safety, potentially by avoiding vascular and/or calcific/thromboembolic complications.
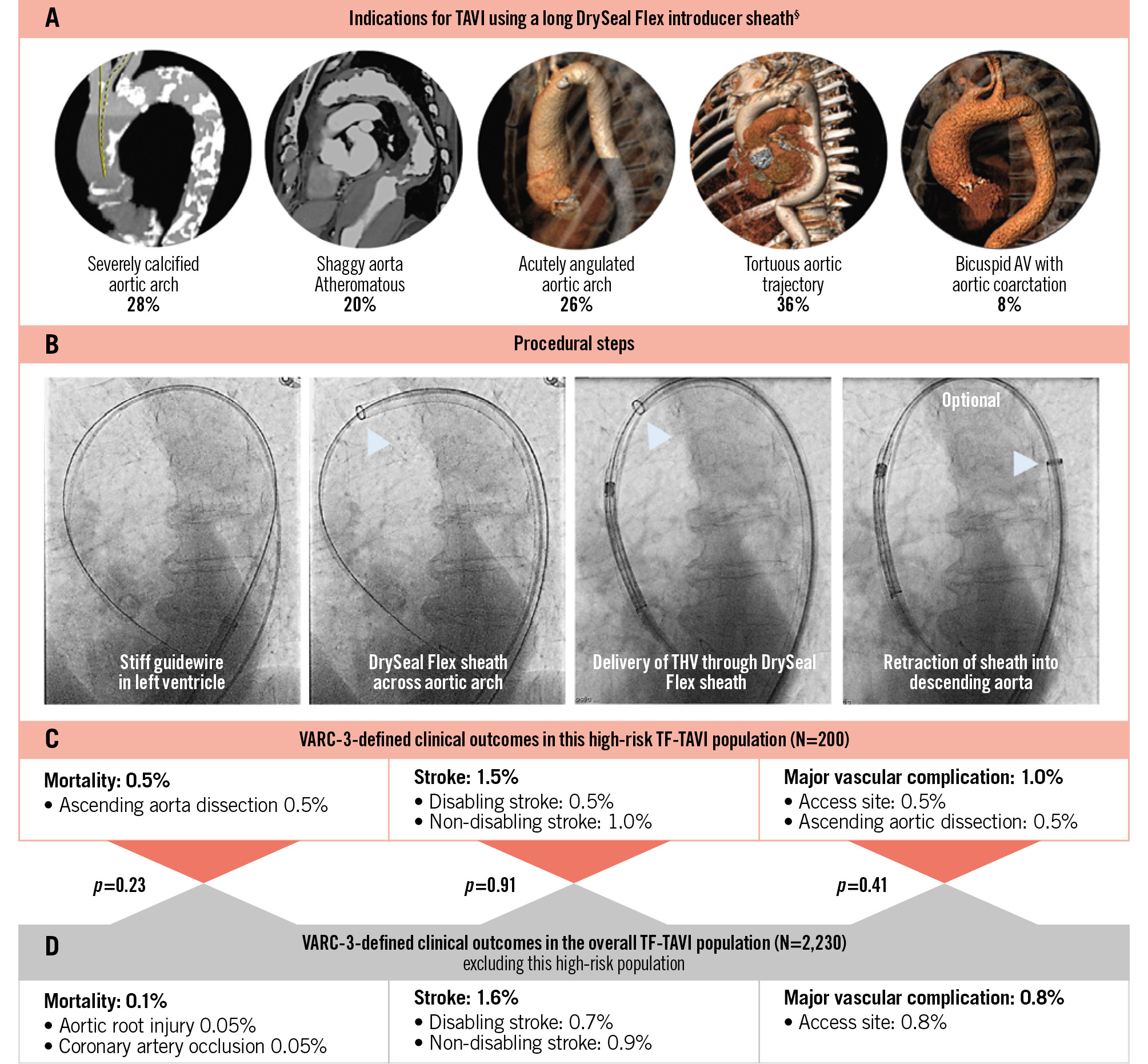
Figure 1. Use of an extended-length introducer sheath for TF-TAVI with self-expanding valves in hostile aortic anatomies. Indications (A), procedural steps (B) and VARC-3-defined clinical outcomes in a patient cohort of 200 patients with a hostile aortic anatomy treated by TF-TAVI using a long DrySeal Flex introducer sheath (C). These outcomes were compared with standard TF-TAVI cases in which a conventional approach was used (D). §One patient can have more than one indication. AV: aortic valve; TAVI: transcatheter aortic valve implantation; TF: transfemoral; THV: transcatheter heart valve; VARC: Valve Academic Research Consortium
Conflict of interest statement
Y. Kobari has received financial support from the Japan Heart Foundation for his fellowship; and speaker fees from Abbott and Boston Scientific. A.A. Khokhar received honoraria from Abbott, Boston Scientific, and Medtronic. G. Bieliauskas received institutional research grants and consulting fees from Boston Scientific. O. De Backer received institutional research grants and consulting fees from Abbott, Boston Scientific, and Medtronic. The other authors have no conflicts of interest to declare relevant to the contents of this paper.
Supplementary data
To read the full content of this article, please download the PDF.
Moving image 1. Use of a long DrySeal Flex introducer sheath for TAVI in higher risk aortic anatomies.

