Cory:
Unlock Your AI Assistant Now!
Abstract
Background: Renal function is a critical factor of ischaemic and bleeding risks in patients with atrial fibrillation (AF) receiving antithrombotic therapy.
Aims: This study aimed to evaluate the impact of renal dysfunction in patients with AF and stable coronary artery disease (CAD) undergoing antithrombotic therapy.
Methods: The Edoxaban Versus Edoxaban With antiPlatelet Agent In Patients With Atrial Fibrillation and Chronic Stable Coronary Artery Disease (EPIC-CAD) trial randomised patients to edoxaban monotherapy or dual antithrombotic therapy (edoxaban plus a single antiplatelet agent). In this prespecified analysis, patients were stratified by creatinine clearance into low (<50 mL/min) or high (≥50 mL/min) groups according to edoxaban dose-reduction criteria. The primary endpoint was net adverse clinical events (NACE: death from any cause, myocardial infarction, stroke, systemic embolism, urgent revascularisation, or major/clinically relevant non-major bleeding) at 12 months.
Results: Of 1,040 randomised patients, 252 (24.2%) had low creatinine clearance; these patients were older and had more comorbidities compared with the 788 patients (75.8%) with high creatinine clearance. Patients with low creatinine clearance experienced higher risks of NACE (hazard ratio [HR] 1.72, 95% confidence interval [CI]: 1.19-2.49; p=0.004), ischaemic events (HR 2.70, 95% CI: 1.09-6.70; p=0.032), and bleeding (HR 1.54, 95% CI: 1.01-2.34; p=0.046). At 12 months, edoxaban monotherapy reduced NACE compared with dual therapy in both the low (12.1% vs 21.7%, HR 0.52, 95% CI: 0.28-0.98; p=0.042) and high creatinine clearance groups (5.2% vs 14.5%, HR 0.40, 95% CI: 0.25-0.65; p<0.001), with no interaction (p for interaction=0.53).
Conclusions: In patients with AF and stable CAD, edoxaban monotherapy led to a lower risk of primary NACE than dual antithrombotic therapy, regardless of renal function. (ClinicalTrials.gov: NCT03718559)
Impaired renal function and the presence of chronic kidney disease (CKD) are well known to markedly increase the risks of both ischaemic and bleeding events12. Therefore, renal function is a crucial factor for the optimal selection of antithrombotic therapy for patients with atrial fibrillation (AF) or coronary artery disease (CAD)34. Given that reduced renal clearance leads to increased drug concentrations and a heightened risk of bleeding events56, dose adjustment of direct oral anticoagulants (DOACs) is essential in patients with AF and impaired renal function. Furthermore, since impaired renal function negatively influences platelet activity, potentially exacerbating the simultaneous risk of bleeding and ischaemic complications7, the presence of renal dysfunction or CKD is a well-known risk factor for high bleeding risk in patients with known CAD8. While choosing antithrombotic therapy for patients with both AF and CAD is challenging910, the task of optimally balancing the risks of ischaemic and bleeding complications becomes much more complicated when impaired renal function is present.
The Edoxaban Versus Edoxaban With antiPlatelet Agent In Patients With Atrial Fibrillation and Chronic Stable Coronary Artery Disease (EPIC-CAD) study was a randomised trial comparing edoxaban monotherapy with dual antithrombotic therapy (edoxaban plus a single antiplatelet agent) in patients with AF and stable CAD11. Given that the EPIC-CAD trial employed a creatinine clearance threshold of 50 mL/min as the criterion for edoxaban dose reduction, it provides a reasonable framework for assessing the impact of renal dysfunction on randomised antithrombotic treatment strategies and their clinical outcomes. Therefore, this prespecified analysis of EPIC-CAD data aims to enhance the interpretation of the trial’s findings based on the status of renal function. Therefore, we examined clinical outcomes according to the randomised antithrombotic strategy (edoxaban monotherapy vs dual antithrombotic therapy) and the category of renal dysfunction in patients with concomitant AF and CAD.
Methods
Study design and patient population
The trial design, methods, and primary outcomes of the EPIC-CAD study have been described previously12. In brief, EPIC-CAD was a multicentre, open-label, adjudicator-masked, randomised trial conducted at 18 hospitals in South Korea between May 2019 and September 2022. A total of 1,040 patients with high-risk AF (CHAâDSâ-VASc score ≥2) and stable CAD (either revascularised or managed medically) were randomly assigned in a 1:1 ratio to receive either edoxaban monotherapy or dual antithrombotic therapy (edoxaban plus a single antiplatelet agent). Stable CAD was defined as one of the following: (1) chronic coronary syndrome treated with percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG) at least 6 months prior to enrolment; (2) acute coronary syndrome treated with PCI or CABG at least 12 months prior to enrolment; or (3) anatomically confirmed CAD (≥50% stenosis of a major epicardial coronary artery on cardiac catheterisation or coronary computed tomography angiography) managed with medical therapy alone. Key exclusion criteria included contraindications for antithrombotic drugs, such as severe coexisting conditions or a high risk of bleeding; a history of intracranial haemorrhage; presence of prosthetic heart valves or moderate-to-severe mitral stenosis; and severe hepatic or renal dysfunction (creatinine clearance <15 mL/min).
The trial was approved by the investigational review board or ethics committee at each participating institution, and all patients provided written informed consent before enrolment. The trial was registered at ClinicalTrials.gov: NCT03718559.
Randomisation, trial regimens, and category of renal dysfunction
Enrolled patients were randomly assigned to receive either standard-dose edoxaban monotherapy (60 mg once daily) or dual antithrombotic therapy, comprising standard-dose edoxaban plus a single antiplatelet agent (either aspirin or a P2Y12 inhibitor, at the discretion of the treating physician), for 12 months. Dose adjustment of edoxaban to 30 mg once daily was indicated in patients with a creatinine clearance of 15-50 mL/min (as calculated with the Cockcroft-Gault formula), a body weight of ≤60 kg, and the use of certain P-glycoprotein inhibitors.
Based on the edoxaban dose-reduction criteria, this prespecified subgroup analysis categorised study participants into low creatinine clearance (<50 mL/min) versus high creatinine clearance (≥50 mL/min) renal function groups. Further analyses within the high creatinine clearance groups distinguished between normal (creatinine clearance of 50-95 mL/min) and extra-normal (creatinine clearance >95 mL/min) levels. Sensitivity analyses, in accordance with the National Kidney Foundation Kidney Disease Outcomes Quality Initiative guidelines13, were also performed using estimated glomerular filtration rate (eGFR), categorising patients into low (eGFR <60 mL/min/1.73 m2, stages 3-4) and normal (eGFR ≥60 mL/min/1.73 m2, stages 1-2) renal function groups.
Outcomes
The primary outcome of the study was net adverse clinical events (i.e., efficacy and safety outcomes), defined as a composite of death from any cause, myocardial infarction (MI), stroke, systemic embolism, unplanned urgent revascularisation, or major/clinically relevant non-major bleeding (as defined by the International Society on Thrombosis and Haemostasis [ISTH])14, at 12 months after randomisation. Secondary efficacy outcomes included the individual components of the primary outcome, a composite of major ischaemic events (death from any cause, MI, ischaemic stroke, or systemic embolism), and a composite of any ischaemic event (death from any cause, MI, ischaemic stroke, systemic embolism, or unplanned urgent revascularisation). Secondary safety outcomes included a composite of major/clinically relevant non-major bleeding, major bleeding, clinically relevant non-major bleeding, and any bleeding event. Prespecified, standard definitions were used to assess clinical outcomes11. All clinical outcomes were adjudicated by an independent clinical events committee, whose members were unaware of the trial-group assignments.
Follow-up assessments were performed at baseline, and at 6 and 12 months after randomisation. At each visit, data on clinical events and concomitant cardiovascular medications were systematically collected. Survival status was cross validated using the Korean National Health Insurance database.
Statistical analysis
The full statistical analysis plan (SAP), including the sample size calculation for EPIC-CAD, has been previously published11. Subgroup analysis by renal function was prespecified in both the trial protocol and the SAP. Baseline characteristics and procedural data were compared between groups using the Student’s t-test for continuous variables and the χ2 or Fisher’s exact test for categorical variables.
For comparisons between creatinine clearance <50 mL/min and ≥50 mL/min, propensity score (PS) matching was performed using a 1:1 nearest-neighbour algorithm with a calliper width of 0.2 of the standard deviation of the logit of the PS, to minimise selection bias and potential confounding. Model discrimination was evaluated using the C-statistic (0.946), and calibration was assessed with the Hosmer-Lemeshow test (χ2=8.177; degrees of freedom=8; p=0.42). Balance between groups was evaluated using standardised mean differences (SMDs), with values <0.1 indicating negligible imbalance. The outcomes were compared by use of Cox proportional hazards regression with robust standard errors that accounted for the clustering of matched pairs.
All analyses were conducted on an intention-to-treat basis. Outcomes of patients randomised to the two antithrombotic strategies (edoxaban monotherapy versus dual antithrombotic therapy) were evaluated according to the creatinine clearance level. Time-to-event estimates for clinical outcomes were obtained using Kaplan-Meier estimates and compared with the log-rank test. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated using Cox proportional hazards models. The proportional hazards assumption was tested for each outcome using Schoenfeld residuals and visual inspection15. Differences in cumulative incidence and corresponding 95% CIs for trial outcomes at 12 months were calculated using Kaplan-Meier estimates and Greenwood standard errors16. Interactions between the randomised antithrombotic strategy and the category of renal function were also tested. Also, the non-linear association between creatinine clearance values (as a continuous variable) and the 12-month predicted probability of adverse outcomes was evaluated using a restricted cubic spline function. Results were visualised by plotting the predicted 12-month probability of adverse outcome versus the creatinine clearance level. No imputation methods were used to infer missing values of baseline variables.
All reported p-values are two-sided. A p-value of <0.05 was considered statistically significant for all tests. No adjustments were made for multiple testing; thus, all findings of the present study must be interpreted as exploratory, given the potential for type I error arising from multiple comparisons. All statistical analyses were performed by independent statisticians using commercially available software (SAS, version 9.4 [SAS Institute] and R, version 4.6.1 [R Foundation for Statistical Computing]).
Results
Study population and baseline characteristics
Between 14 May 2019 and 19 September 2022, a total of 1,040 patients were enrolled in the EPIC-CAD trial across 18 sites in South Korea. Of the 1,040 patients randomised in the EPIC-CAD trial, 252 patients (24.2%) had low creatinine clearance (<50 mL/min), and 788 patients (75.8%) had high creatinine clearance (≥50 mL/min). Among the patients with low creatinine clearance, 128 (50.8%) were assigned to edoxaban monotherapy and 124 (49.2%) to dual antithrombotic therapy. In the high creatinine clearance group, 396 patients (50.3%) were assigned to edoxaban monotherapy and 392 (49.7%) to dual antithrombotic therapy (Figure 1).
Baseline characteristics of patients according to the levels of creatinine clearance are presented in Table 1 and Supplementary Table 1. Compared with patients with high creatinine clearance (≥50 mL/min), those with low creatinine clearance (<50 mL/min) were older, more likely to be female, had a lower body weight, exhibited a higher prevalence of clinical comorbidities, and had higher mean CHA2DS2-VASc (5.4±1.5 vs 4.0±1.4) and HAS-BLED scores (2.4±0.8 vs 2.1±0.8). Within each creatinine clearance subgroup, the baseline characteristics of patients were mostly well balanced between the randomised antithrombotic strategies of edoxaban monotherapy and dual antithrombotic therapy (Supplementary Table 2).
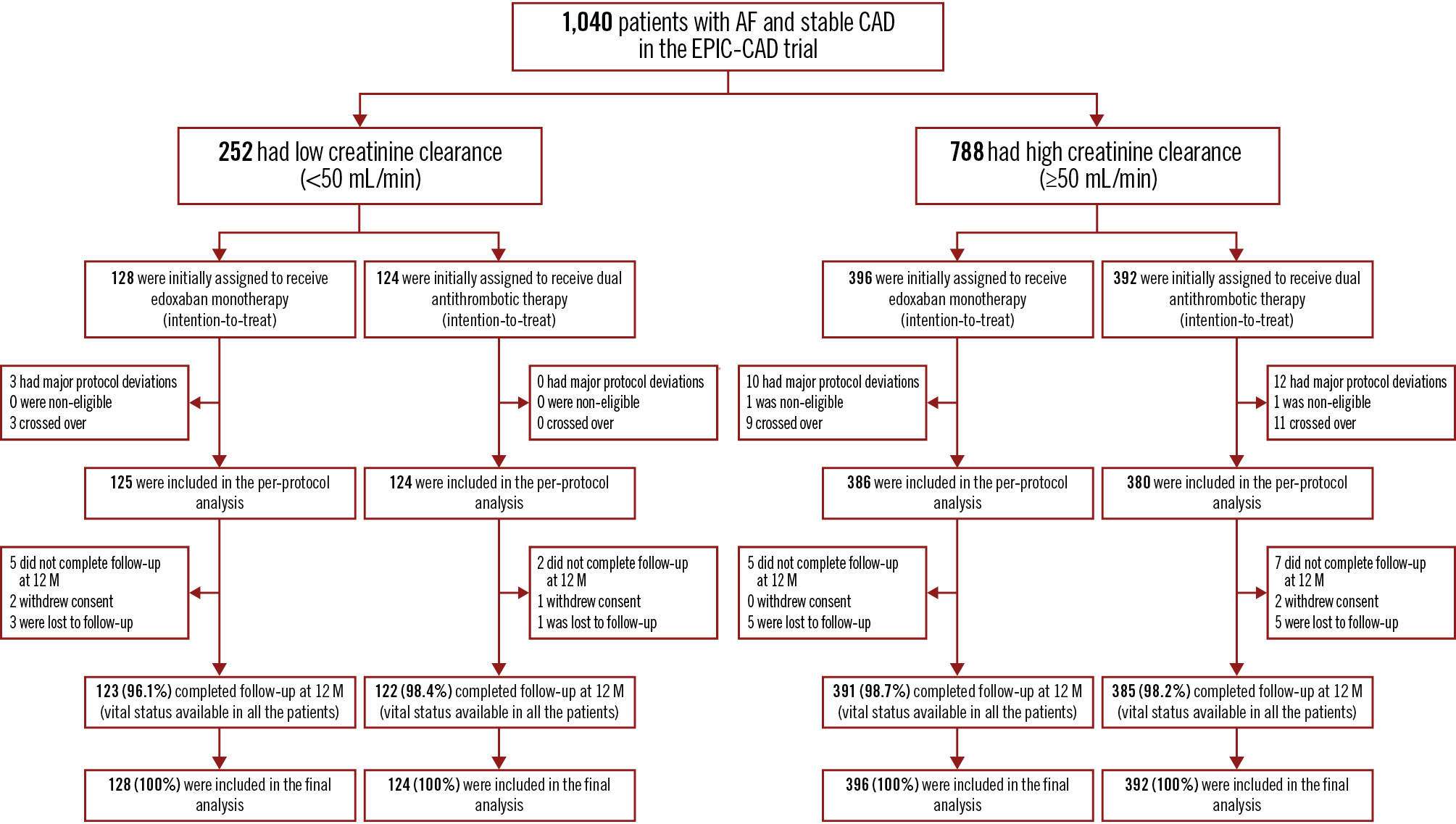
Figure 1. Enrolment, randomisation, and follow-up stratified by the status of renal function. Renal function was stratified into low (<50 mL/min) or high (≥50 mL/min) creatinine clearance groups. Patients were randomly assigned in a 1:1 ratio to receive edoxaban monotherapy or dual antithrombotic therapy consisting of edoxaban plus a single antiplatelet agent (either aspirin or a P2Y12 inhibitor), and propensity score matching was performed to minimise baseline imbalances. Propensity-matched groups were generated based on clinically relevant baseline characteristics, including age, sex, body weight, comorbidities (diabetes, hypertension, hyperlipidaemia, heart failure), cardiovascular history, and concomitant medications. AF: atrial fibrillation; CAD: coronary artery disease; EPIC-CAD: Edoxaban Versus Edoxaban With antiPlatelet Agent In Patients With Atrial Fibrillation and Chronic Stable Coronary Artery Disease
Table 1. Baseline characteristics of patients stratified by renal function status.
| Baseline characteristics | Before PS matching | After PS matchingd | |||||
|---|---|---|---|---|---|---|---|
| Low creatinine clearance (n=252) | High creatinine clearance (n=788) | p-value | SMD | Low creatinine clearance (n=99) | High creatinine clearance (n=99) | SMD | |
| Age, years | 78.7±6.3 | 70.0±7.6 | <0.001 | 1.247 | 75.7±6.2 | 76.2±6.3 | 0.074 |
| Male sex | 158 (62.7) | 644 (81.7) | <0.001 | 0.435 | 58 (58.6) | 55 (55.6) | 0.061 |
| Weight, kg | 62.1±10.3 | 70.8±10.8 | <0.001 | 0.822 | 60.3±9.6 | 60.3±10.4 | 0.002 |
| Body mass indexa, kg/m² | 24.1±3.2 | 25.8±3.2 | <0.001 | 0.515 | 23.4±3.2 | 23.9±3.8 | 0.139 |
| Diabetes mellitus | 120 (47.6) | 301 (38.2) | 0.008 | 0.191 | 37 (37.4) | 34 (34.3) | 0.063 |
| Hypertension | 206 (81.7) | 639 (81.1) | 0.82 | 0.017 | 80 (80.8) | 80 (80.8) | <0.001 |
| Hyperlipidaemia or statin use | 235 (93.3) | 737 (93.5) | 0.88 | 0.011 | 90 (90.9) | 91 (91.9) | 0.036 |
| Current smoker | 13 (5.2) | 74 (9.4) | 0.04 | 0.164 | 6 (6.1) | 5 (5.1) | 0.044 |
| Previous myocardial infarction | 48 (19.0) | 123 (15.6) | 0.20 | 0.091 | 12 (12.1) | 11 (11.1) | 0.032 |
| Congestive heart failure | 65 (25.8) | 140 (17.8) | 0.005 | 0.195 | 19 (19.2) | 21 (21.2) | 0.050 |
| History of cerebrovascular disease | 56 (22.2) | 98 (12.4) | <0.001 | 0.261 | 15 (15.2) | 15 (15.2) | <0.001 |
| History of peripheral artery disease | 28 (11.1) | 50 (6.3) | 0.01 | 0.170 | 4 (4.0) | 5 (5.1) | 0.049 |
| Creatinine clearance, mL/minb | 40.4±7.3 | 74.9±19.1 | <0.001 | 2.387 | 42.0±6.4 | 64.6±15.8 | 1.879 |
| Type of atrial fibrillation | 0.63 | 0.035 | 0.081 | ||||
| Paroxysmal | 136 (54.0) | 439 (55.7) | 55 (55.6) | 51 (51.5) | |||
| Persistent or permanent | 116 (46.0) | 349 (44.3) | 44 (44.4) | 48 (48.5) | |||
| CHA2DS2-VASc scorec | <0.001 | 1.038 | 0.029 | ||||
| Mean | 5.4±1.5 | 4.0±1.4 | 4.8±1.4 | 4.8±1.4 | |||
| Median | 6 (4-6) | 4 (3-5) | 5 (4-6) | 5 (4-6) | |||
| Actual dose of edoxaban | <0.001 | 1.471 | 0.048 | ||||
| 60 mg | 33 (13.1) | 565 (71.7) | 23 (23.2) | 21 (21.2) | |||
| 30 mg | 219 (86.9) | 223 (28.3) | 79 (79.8) | 81 (81.8) | |||
| Values are mean±SD, n (%), or median (IQR). Percentages may not add up to 100% due to rounding. aThe body mass index is the weight in kilograms divided by the square of the height in metres. bCreatinine clearance was assessed using the Cockcroft-Gault formula. cThe CHA2DS2-VASc score is a clinical tool used to assess the risk of stroke among people with atrial fibrillation. Scores are weighted based on the presence of congestive heart failure, hypertension, diabetes mellitus, and vascular disease; a history of stroke or transient ischaemic attack; age (65-74 years or ≥75 years); and sex. Scores range from 0 to 9, with higher scores indicating a greater risk28. dPropensity-matched groups were generated based on clinically relevant baseline characteristics, including age, sex, body weight, comorbidities (diabetes, hypertension, hyperlipidaemia, heart failure), cardiovascular history, and concomitant medication. IQR: interquartile range; PS: propensity score; SD: standard deviation; SMD: standardised mean difference | |||||||
Treatment and follow-up
Details regarding the antithrombotic regimens administered before or after randomisation are summarised in Supplementary Table 3. Before randomisation, 47.2% of the patients in the low creatinine clearance group received dual antithrombotic therapy, 44.4% received oral anticoagulants alone, and 7.5% received a single antiplatelet agent. This pattern was similar for patients in the high creatinine clearance group. After randomisation, most patients started their assigned antithrombotic treatment, irrespective of the creatinine clearance category. Details regarding trial regimens and concomitant cardioactive medication use at randomisation and during follow-up are provided in Supplementary Table 4.
Clinical outcomes
By creatinine clearance levels
Primary and secondary outcomes were assessed at 12 months in 98.2% of all patients (Figure 1). Data regarding vital status were obtained for all patients. Crude and adjusted clinical outcomes stratified by the level of creatinine clearance are summarised in Table 2, Supplementary Table 5, and Figure 2. As expected, the incidence of the primary net adverse clinical events at 12 months was significantly higher in the low creatinine clearance group than in the high creatinine clearance group in both the crude and PS-matched analyses (Figure 2). However, the cumulative incidence of major ischaemic events at 12 months was significantly higher in the low creatinine clearance group than in the high creatinine clearance group in the crude analysis, whereas this difference was no longer significant in the PS-matched analysis. The incidence of major/clinically relevant non-major bleeding was higher in the low creatinine clearance group. Major bleeding events were also more frequent in the low creatinine clearance group than in the high creatinine clearance group.
Table 2. Primary and key secondary outcomes at 1 year in patients stratified by creatinine clearance of 50 mL/min.
| Outcomes | Before PS matching | After PS matching | ||||||
|---|---|---|---|---|---|---|---|---|
| Low creatinine clearance (n=252) | High creatinine clearance (n=788) | HR (95% CI) | p-value | Low creatinine clearance (n=99) | High creatinine clearance (n=99) | HR (95% CI) | p-value | |
| Net adverse clinical eventsa | 39 (17.0) | 74 (9.8) | 1.72 (1.19-2.49) | 0.004 | 17 (18.5) | 4 (4.8) | 3.53 (1.24-10.07) | 0.018 |
| Composite major ischaemic eventsb | 8 (3.5) | 8 (1.1) | 2.70 (1.09-6.70) | 0.032 | 2 (2.2) | 1 (1.0) | 2.02 (0.19-21.99) | 0.564 |
| Major bleeding or clinically relevant non-major bleeding | 30 (13.3) | 63 (8.2) | 1.54 (1.01-2.34) | 0.046 | 13 (14.4) | 2 (2.0) | 4.37 (1.16-16.43) | 0.029 |
| Major bleeding | 13 (5.7) | 15 (2.0) | 2.80 (1.37-5.74) | 0.005 | 7 (8.2) | 0 (0) | NE | - |
| Values are the number of patients with events (estimated percentages), calculated using Kaplan-Meier survival analysis of data from the intention-to-treat population; therefore, the reported percentages may not reflect the ratio of the numerator and the denominator. The 95% confidence intervals for secondary outcomes were not adjusted for multiple comparisons; consequently, inferences drawn from these intervals may not be reproducible. Kaplan-Meier event rates and Cox proportional hazard ratios were adjusted for age, sex, weight, diabetes mellitus, current smoking status, congestive heart failure, cerebrovascular disease, peripheral artery disease, and prior coronary revascularisation. aNet adverse clinical events were defined as a composite of death from any cause, myocardial infarction, stroke, systemic thromboembolic event, unplanned urgent revascularisation, or major/clinically relevant non-major bleeding (according to the International Society on Thrombosis and Haemostasis definition). A p-value<0.001 indicated the superiority of edoxaban monotherapy over dual antithrombotic therapy. bThe composite of major ischaemic events was defined as a composite of death from any cause, myocardial infarction, ischaemic stroke, or systemic embolism. CI: confidence interval; HR: hazard ratio; NE: not estimable; PS: propensity score | ||||||||
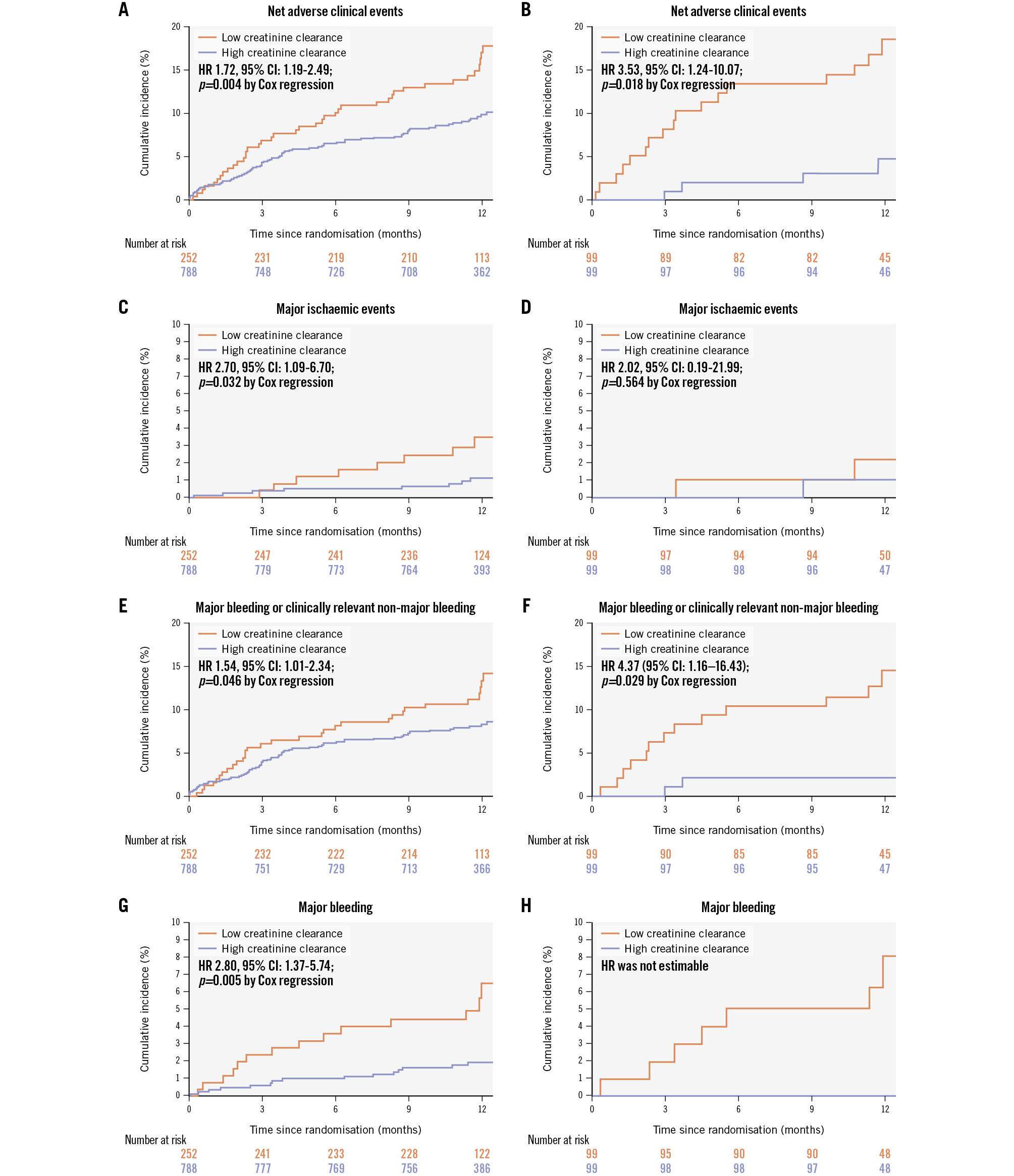
Figure 2. Time-to-event curves for clinical outcomes stratified by creatinine clearance of 50 mL/min. Renal function was stratified into low (<50 mL/min) or high (≥50 mL/min) creatinine clearance groups. Primary outcome refers to the net adverse outcome, which includes death from any cause, myocardial infarction, stroke, systemic embolism, unplanned emergency procedures, and major/clinically significant non-major bleeding. Secondary outcomes included major ischaemic events, major/clinically significant non-major bleeding, and major bleeding. The percentages for events were calculated using Kaplan-Meier estimates and compared with Cox regression tests. Propensity-matched groups were generated based on clinically relevant baseline characteristics, including age, sex, body weight, comorbidities (diabetes, hypertension, hyperlipidaemia, heart failure), cardiovascular history, and concomitant medications. A, C, E, G) Results from the unmatched cohort; (B, D, F, H) outcomes after propensity score matching. CI: confidence interval; HR: hazard ratio
By randomised antithrombotic strategy and renal function
The primary and secondary outcomes according to the randomised antithrombotic strategy, stratified by creatinine clearance levels, are presented in Table 3, Supplementary Table 6, and Figure 3. In the low creatinine clearance group, the incidence of the primary net adverse events at 12 months was significantly lower with edoxaban monotherapy than with dual antithrombotic therapy (12.1% vs 21.7%, respectively; HR 0.52, 95% CI: 0.28-0.98; p=0.04). Similarly, in the high creatinine clearance group, the incidence of the primary net adverse events was also lower with edoxaban monotherapy compared to dual antithrombotic therapy (5.2% vs 14.5%; HR 0.40, 95% CI: 0.25-0.65; p<0.001). Thus, no significant interaction was observed between the randomised antithrombotic strategy and the creatinine clearance category (p for interaction=0.53) (Central illustration).
In both groups of low or high creatinine clearance category, the cumulative incidence of major or any ischaemic events did not significantly differ between the edoxaban monotherapy and dual antithrombotic therapy groups. Regardless of creatinine clearance levels, the cumulative incidence of major/clinically relevant non-major bleeding at 12 months was significantly lower with edoxaban monotherapy than with dual antithrombotic therapy. The cumulative incidence of major bleeding at 12 months was significantly lower with edoxaban monotherapy than with dual antithrombotic therapy in the low creatinine clearance group; however, this trend was not significant in the high creatinine clearance group (Figure 3). Detailed information regarding the severity of bleeding, stratified by bleeding criteria and sites, is summarised in Supplementary Table 7. No significant interaction was observed between the randomised antithrombotic strategy and the creatinine clearance category with respect to major ischaemic events (p for interaction=0.65), any ischaemic event (p for interaction=0.69), major/clinically relevant non-major bleeding (p for interaction=0.78), and major bleeding (p for interaction=0.30).
Table 3. Primary and key secondary outcomes at 1 year in patients stratified by creatinine clearance and randomisation strategy.
| Outcomes | Low creatinine clearance (n=252) | High creatinine clearance (n=788) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Edoxaban monotherapy (n=128) | Dual antithrombotic therapy (n=124) | HR (95% CI) | p-value | Edoxaban monotherapy (n=396) | Dual antithrombotic therapy (n=392) | HR (95% CI) | p-value | p-int | |
| Net adverse clinical eventsa | 14 (12.1) | 25 (21.7) | 0.52 (0.28-0.98) | 0.04 | 20 (5.2) | 54 (14.5) | 0.40 (0.25-0.65) | <0.001 | 0.53 |
| Composite major ischaemic eventsb | 4 (3.3) | 4 (3.6) | 1.00 (0.26-3.90) | >0.99 | 4 (1.1) | 4 (1.2) | 1.54 (0.43-5.50) | 0.50 | 0.65 |
| Major bleeding or clinically relevant non-major bleeding | 8 (7.3) | 22 (19.2) | 0.37 (0.17-0.79) | 0.01 | 15 (3.9) | 48 (12.7) | 0.32 (0.18-0.55) | <0.001 | 0.78 |
| Major bleeding | 2 (2.4) | 11 (9.2) | 0.17 (0.04-0.76) | 0.02 | 4 (1.0) | 11 (3.0) | 0.44 (0.15-1.28) | 0.133 | 0.30 |
| Values are the number of patients with events (estimated percentages), calculated using Kaplan-Meier survival analysis of data from the intention-to-treat population; therefore, the reported percentages may not reflect the ratio of the numerator and the denominator. The 95% confidence intervals for secondary outcomes were not adjusted for multiple comparisons; consequently, inferences drawn from these intervals may not be reproducible. aNet adverse clinical events were defined as a composite of death from any cause, myocardial infarction, stroke, systemic thromboembolic event, unplanned urgent revascularisation, or major/clinically relevant non-major bleeding (according to the International Society on Thrombosis and Haemostasis definition). A p-value of <0.001 indicated the superiority of edoxaban monotherapy over dual antithrombotic therapy. bThe composite of major ischaemic events was defined as the occurrence of any of the following: death from any cause, myocardial infarction, ischaemic stroke, or systemic embolism. CI: confidence interval; HR: hazard ratio; p-int: p for interaction | |||||||||
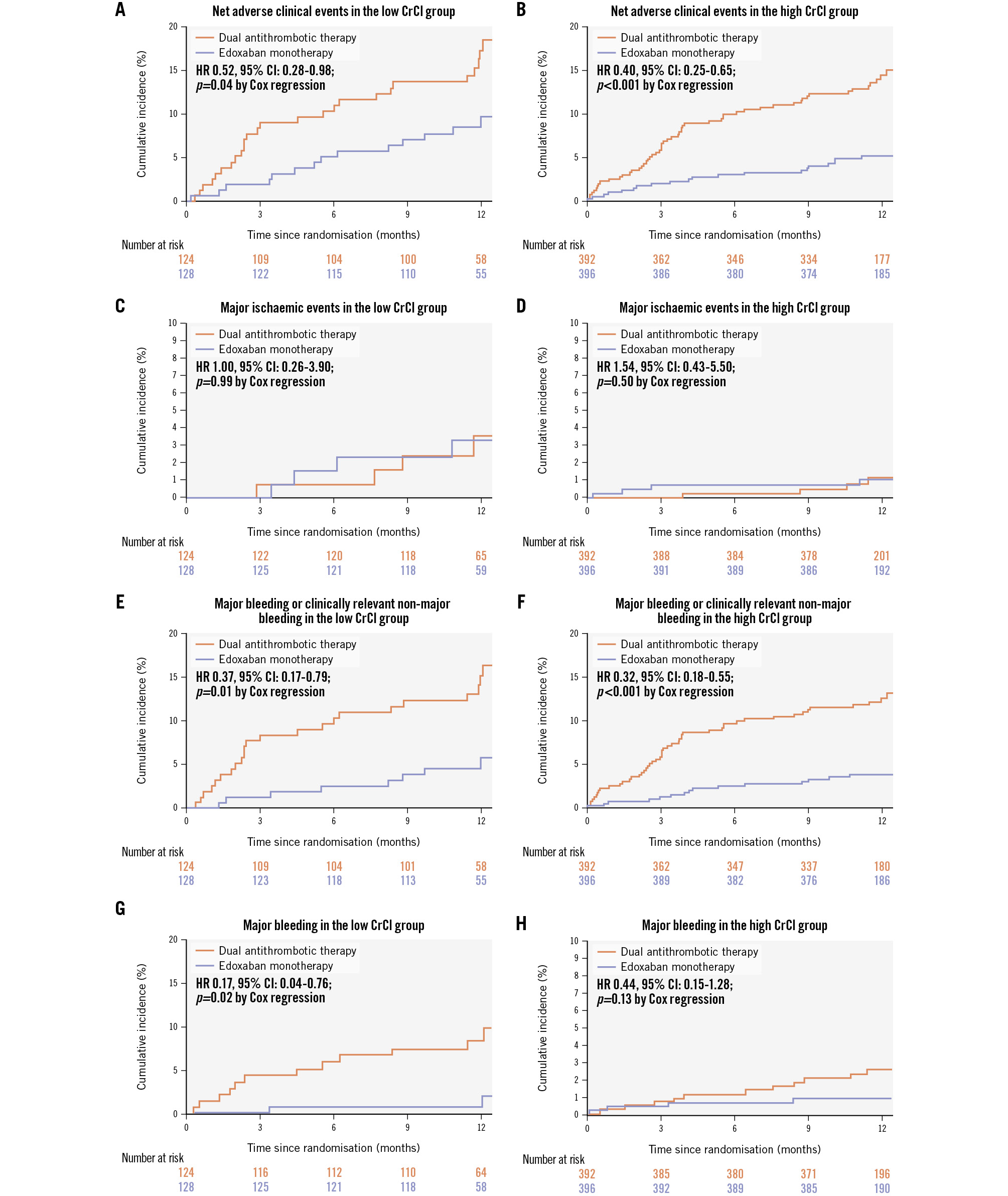
Figure 3. Time-to-event curves for the primary composite outcome stratified by creatinine clearance level and randomised follow-up strategy. Renal function was stratified into low (<50 mL/min) or high (≥50 mL/min) creatinine clearance groups. Primary outcome refers to the net adverse outcome, which includes death from any cause, myocardial infarction, stroke, systemic embolism, unplanned emergency procedures, and major/clinically significant non-major bleeding. Secondary outcomes included major ischaemic events, major/clinically significant non-major bleeding, and major bleeding. The percentages of events were calculated using Kaplan-Meier estimates and were compared with Cox regression tests. A, C, E, G) Results from the low creatinine clearance groups; (B, D, F, H) outcomes from the high creatinine clearance groups. CI: confidence interval; CrCl: creatinine clearance; HR: hazard ratio
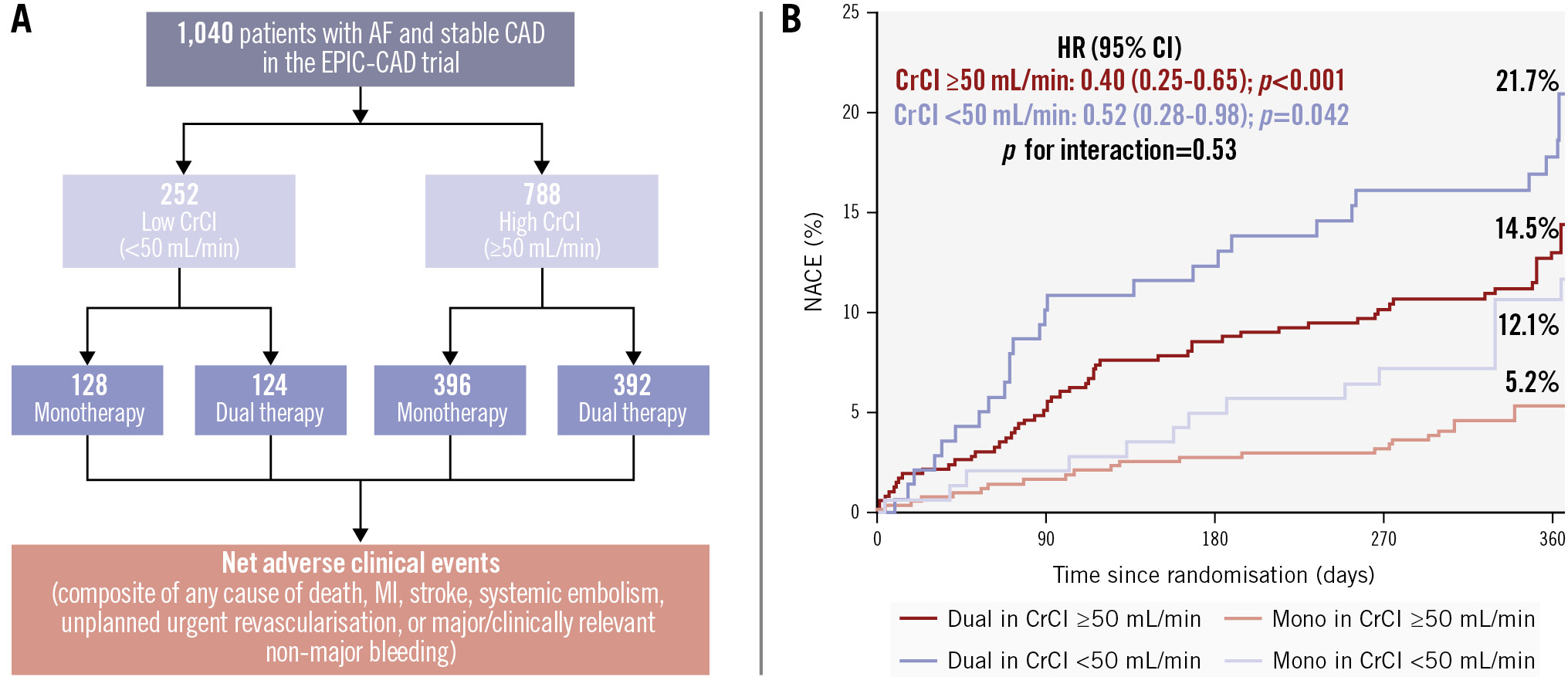
Central illustration. Impact of creatinine clearance on the antithrombotic therapeutic strategy in patients with AF and stable CAD: findings from the EPIC-CAD trial. A) Patient distribution in the study; (B) time-to-event curves for the incidence of NACE according to dual or monotherapy and high or low creatinine clearance. AF: atrial fibrillation; CAD: coronary artery disease; CI: confidence interval; CrCl: creatinine clearance; EPIC-CAD: Edoxaban Versus Edoxaban With antiPlatelet Agent In Patients With Atrial Fibrillation and Chronic Stable Coronary Artery Disease; HR: hazard ratio; MI: myocardial infarction; NACE: net adverse clinical events
Sensitivity analyses
The outcomes of analyses performed, stratified by normal (creatinine clearance 50-95 mL/min) and extra-normal (creatinine clearance >95 mL/min) levels, are summarised and illustrated in Supplementary Table 8 and Supplementary Figure 1, respectively. In patients with creatinine clearance of 50-95 mL/min, the incidence of primary net adverse events, major bleeding, or clinically relevant non-major bleeding was significantly lower with edoxaban monotherapy compared with dual antithrombotic therapy. However, this difference was not observed in patients with creatinine clearance >95 mL/min. Sensitivity analyses using an eGFR cutoff of 60 mL/min/1.73 m2 showed results that were consistent with the overall findings (Supplementary Table 9, Supplementary Figure 2). In a sensitivity analysis restricted to patients with high creatinine clearance and receiving a 60 mg dose of edoxaban, the results were consistent with the primary findings (Supplementary Table 10): edoxaban monotherapy was associated with a lower risk of the primary net adverse clinical events, major bleeding, or clinically relevant non-major bleeding compared with dual antithrombotic therapy. Using a linear model, the probability of the primary outcome substantially increased with lower baseline creatinine clearance, and this association was consistent across each stratum of the edoxaban monotherapy and dual antithrombotic therapy groups, with no significant interaction (p for interaction=0.37) (Supplementary Figure 3).
Discussion
In this prespecified secondary analysis of the EPIC-CAD trial, we evaluated the comparative outcomes of different antithrombotic strategies (edoxaban monotherapy vs dual antithrombotic therapy) in patients with high-risk AF and stable CAD according to their renal function status. The key findings are summarised as follows: (1) patients with lower creatinine clearance were older, had a lower body weight, and had a higher risk of clinical comorbidities associated with an increased risk of ischaemic and bleeding events; (2) the incidences of primary net adverse events, major/clinically relevant bleeding events, and major bleeding events were generally higher in patients with low creatinine clearance than in those with high creatinine clearance; (3) the 12-month rate of primary net adverse events was significantly lower with edoxaban monotherapy than with dual antithrombotic therapy in both the low and high creatinine clearance groups (Central illustration). This observation was driven mainly by a lower incidence of bleeding events. Despite the low number of ischaemic outcomes, the incidences of ischaemic events and mortality were similar in the randomised antithrombotic groups; and (4) overall findings were consistent across various categories of renal function.
In contemporary clinical practice, renal function is a key determinant in selecting optimal antithrombotic therapy due to the increased risks of both ischaemic and bleeding events in patients with AF, CAD, or both conditions121718. Such detrimental effects of renal dysfunction on ischaemic or bleeding complications were also evident in the current analysis of the EPIC-CAD trial. Also, we observed that edoxaban monotherapy was associated with a lower incidence of the primary net adverse clinical events compared with dual antithrombotic therapy in patients with either reduced or normal creatinine clearance. This key finding was mainly driven by a lower incidence of clinically relevant bleeding events without a corresponding increase in ischaemic events. Prior studies have reported that DOAC underdosing was associated with an increased risk of ischaemic events in patients with reduced renal function (i.e., low creatinine clearance), despite a lower incidence of bleeding events19202122. In the present study, the creatinine clearance cutoff of <50 mL/min was prespecified in this analysis and corresponds to the approved criterion for edoxaban dose reduction. Although the creatinine clearance cutoff of <50 mL/min reflects a pharmacokinetic threshold for edoxaban dose adjustment rather than a conventional prognostic boundary, sensitivity analyses using eGFR <60 mL/min/1.73 m2 yielded consistent results, supporting the robustness of our findings. From a clinical viewpoint, our findings suggest that edoxaban monotherapy was associated with a lower risk of primary net adverse clinical events and bleeding events compared with dual antithrombotic therapy, irrespective of baseline renal function. While no statistically significant difference in ischaemic events was observed between the treatment strategies, these ischaemic comparisons should be considered exploratory given the low event rates and limited statistical power.
A substudy of the Atrial Fibrillation and Ischemic Events with Rivaroxaban in Patients With Stable Coronary Artery Disease Study (AFIRE), which used a similar eGFR cutoff value of 50 mL/min, reported that impaired renal function (eGFR <50 mL/min) was associated with bleeding events when recurrent events were considered. Moreover, the bleeding risk remained high over time in patients with decreased renal function23. However, comparative outcomes of randomised antithrombotic strategies stratified by renal function were not reported. In the current study, the detrimental effect of renal dysfunction on bleeding events and net adverse clinical events was more remarkable. Furthermore, the superior effect of edoxaban monotherapy over dual antithrombotic therapy with regard to primary net adverse events and clinically relevant bleeding events was consistent across patients with or without decreased renal function. From a clinical perspective, these findings can directly inform decision-making regarding optimal antithrombotic therapy in patients with high-risk AF and concomitant CAD, especially those with reduced kidney function.
Interestingly, previous reports have suggested that patients with creatinine clearance >95 mL/min may exhibit decreased plasma concentrations of edoxaban, which potentially reduce the drug efficacy2425. In an explorative analysis of the Effective Anticoagulation With Factor Xa Next Generation in Atrial Fibrillation-Thrombolysis in Myocardial Infarction 48 (ENGAGE AF-TIMI 48) trial26, the relative efficacy of higher-dose edoxaban in preventing thromboembolic events, compared with warfarin, was lower in patients with creatinine clearance >95 mL/min. Nevertheless, the primary net clinical outcome remained favourable for higher-dose edoxaban, with persistently lower rates of major bleeding than warfarin.
Limitations
The present study has several limitations. First, as a subgroup analysis of a prospective randomised controlled trial, the present study was limited by a relatively small sample size and reduced statistical power. Accordingly, the findings should be considered exploratory, and further prospective studies are warranted to confirm these observations. Second, although patients with creatinine clearance <50 mL/min were a prespecified subgroup of interest for the original EPIC-CAD trial, no adjustment for multiple testing was made; therefore, the findings should not be used to infer definitive treatment effects. Third, randomisation in the EPIC-CAD trial was not stratified by baseline renal function. Although baseline characteristics were generally balanced between the randomised treatment groups within the creatinine clearance strata, residual confounding due to unmeasured factors may remain in this subgroup analysis. Accordingly, treatment effects may be partly confounded despite multivariable adjustment. Fourth, renal function was assessed only at baseline in the present analysis, and serial reassessment of creatinine clearance during follow-up was not available. Given the dynamic nature of renal function in elderly and comorbid patients with AF, some degree of misclassification may have occurred, which could attenuate the observed differences between the treatment strategies. Lastly, the present study was conducted exclusively in an East Asian population, who are known to exhibit a higher bleeding risk phenotype and distinct pharmacodynamic responses to DOACs compared with Western populations, a phenomenon often referred to as the “East Asian paradox”27. These ethnic and demographic differences, including lower body weight and a different balance between ischaemic and bleeding risks, may influence both the safety and efficacy profiles of antithrombotic strategies. Accordingly, the generalisability of our findings to Western populations should be interpreted with caution, and further studies in more diverse, non-East Asian cohorts are warranted to confirm the external validity of these observations.
Conclusions
In this prespecified secondary analysis of the EPIC-CAD trial, impaired renal function was significantly associated with an increased risk of primary net adverse clinical events and major/clinically relevant bleeding events. Nevertheless, the benefit of edoxaban monotherapy over dual antithrombotic therapy, including reductions in the primary net outcome and bleeding events, was consistent irrespective of renal function status, with no evidence of an excess in ischaemic events.
Impact on daily practice
Patients with atrial fibrillation and stable coronary artery disease who have impaired renal function face substantially higher risks of bleeding and net adverse clinical events. In this analysis, edoxaban monotherapy consistently reduced net adverse outcomes compared with dual antithrombotic therapy across all levels of renal function, without increasing ischaemic events. These findings support a monotherapy-first approach as a practical and safe antithrombotic strategy even in patients with reduced kidney function.
Acknowledgements
We thank the staff members of the EPIC-CAD trial, the members of the cardiology departments at the participating centres, and the study coordinators for their efforts in collecting clinical data and ensuring their accuracy and completeness.
Funding
This study was supported by the Cardiovascular Research Foundation, Daiichi Sankyo, and Daewoong Pharmaceutical.
Conflict of interest statement
D.-W. Park has received research grants from the Cardiovascular Research Foundation, Daewoong Pharmaceutical, and Daiichi Sankyo. G.-B. Nam has received research grants from the Cardiovascular Research Foundation, Daewoong Pharmaceutical, and Daiichi Sankyo. The other authors have no conflicts of interest to declare.
Supplementary data
To read the full content of this article, please download the PDF.

