Cory:
Unlock Your AI Assistant Now!
Abstract
Background: The recurrences of acute coronary syndrome (ACS) and target vessel failure after percutaneous coronary intervention (PCI) remain clinical concerns. Intravascular imaging, such as optical coherence tomography (OCT) or intravascular ultrasound (IVUS), has demonstrated clinical benefits in patients with stable coronary disease; however, the benefits of its use remains unclear in patients with ACS.
Aims: This study aimed to investigate the benefit of imaging-guided PCI in patients with ACS on the recurrence of ACS using a nationwide database in Japan.
Methods: This retrospective observational study used records from the National Database between April 2014 and March 2021. We included patients hospitalised with ACS aged ≥20 years who had undergone first-time PCI and divided them into imaging-guided PCI (OCT or IVUS) and angiography-guided PCI groups. The primary outcome was ACS recurrence during a 3-year follow-up period. We analysed the association between intravascular imaging and the outcome using inverse probability of treatment weighting.
Results: Among the patients with ACS, angiography-guided PCI, OCT-guided PCI, and IVUS-guided PCI were performed in 32,044, 22,748, and 297,944 patients, respectively. During the study period, both OCT- and IVUS-guided PCI rates increased, from 4.7% to 6.9% and from 77.0% to 87.9%, respectively. OCT-guided PCI was associated with a lower risk of ACS recurrence (hazard ratio [HR] 0.81, 95% confidence interval [CI]: 0.71-0.91; p<0.001); IVUS-guided PCI was also associated with a lower risk of ACS recurrence (HR 0.76, 95% CI: 0.71-0.82; p<0.001).
Conclusions: In real-world clinical practice, the rates of both OCT- and IVUS-guided PCI have increased and have been associated with a lower risk of ACS recurrence compared with angiography-guided PCI in patients with ACS.
Percutaneous coronary intervention (PCI) for patients with acute coronary syndrome (ACS) is now widely performed and has reduced acute-phase mortality. However, recurrent ACS and target vessel failure remain clinical concerns. A previous study reported that approximately 7% of ACS patients developed target vessel failure within 1 year1. These chronic-phase coronary events frequently occur in patients with complex lesions, including culprit lesions with residual lipid-rich plaque, bifurcation lesions, and heavily calcified lesions234.
Intravascular imaging, including optical coherence tomography (OCT) and intravascular ultrasound (IVUS), has emerged as an effective modality to enhance culprit lesion assessment and procedural accuracy. Imaging-guided PCI improves lesion characterisation; enables precise stent landing, sizing, and implantation; and helps detect complications such as malapposition or edge dissection that are often missed with angiography guidance alone5678. The use of intravascular imaging in patients with chronic coronary syndrome is a Class 1 recommendation for the assessment of procedural risks and postprocedural outcomes, and has been associated with improved procedure results, including stent underexpansion and a lower occurrence of unplanned PCI91011121314151617.
Following the guideline recommendations for chronic coronary syndrome, the use of intravascular imaging in patients with ACS with complex lesions became a Class 1 recommendation in the current, updated guidelines1. A recent large-scale randomised controlled trial (RCT) reported that IVUS-guided PCI for ACS reduced target vessel failure compared with angiography-guided PCI1. However, generalisable evidence supporting both IVUS- and OCT-guided PCI in real-world settings remains limited. Notably, evidence regarding OCT-guided PCI in patients with ACS is limited1819202122.
Therefore, we aimed to investigate the association of OCT- and IVUS-guided PCI in ACS patients with recurrence of ACS using the nationwide administrative database in Japan.
Methods
Study design and database
This retrospective, observational study analysed data from the National Database of Health Insurance Claims and Specific Health Checkups of Japan (NDB) spanning from April 2014 to March 2021. The NDB provides anonymised individual-level data on diagnoses, procedures, prescriptions, and medical devices in both inpatient and outpatient settings. It does not include laboratory results. A patient-matching approach allows long-term follow-up, enabling the tracking of over 90% of patients23. Coding details are provided in Supplementary Table 1.
Study population
We extracted data on patients aged 20 years or older who had a first-time ACS and underwent PCI. We excluded patients who (1) had no insurance data for >365 days before hospitalisation, (2) died or developed ACS after PCI during hospitalisation, (3) were hospitalised for more than 30 days, (4) required mechanical circulatory support, (5) developed ventricular fibrillation or cardiopulmonary arrest, or (6) underwent coronary artery bypass grafting during hospitalisation. We collected data on patient age, sex, comorbidities, medical history, prescriptions before and during hospitalisation, fiscal year, emergency admission, and PCI procedure. Medications before admission were defined as prescriptions recorded within a period of 90 days prior to first-time PCI. Comorbidities and medical histories were identified by reviewing diagnoses from the 365 days before hospitalisation. Patients were divided into two groups according to whether the index PCI was performed with or without intravascular imaging: an imaging-guided PCI (OCT- or IVUS-guided PCI) group and an angiography-guided PCI group (Supplementary Figure 1).
Outcomes
The primary outcome was ACS recurrence after discharge, and the secondary outcome was a composite of all-cause death and ACS recurrence. The results were analysed over a 3-year follow-up period. Event-free censoring was based on the occurrence of the last medical claim during the study period.
Statistical analysis
The patients’ baseline clinical and procedural characteristics are reported as numerical values and percentages for categorical variables. We described the annual changes in the utilisation rate of intravascular imaging. We set up two comparisons: (i) the imaging-guided PCI group versus the angiography-guided PCI group, and (ii) the OCT- and IVUS-guided PCI groups versus the angiography-guided PCI group. We generated a Kaplan-Meier curve and performed a univariable Cox regression analysis to compare the imaging-guided PCI group (the OCT- and IVUS-guided PCI groups) with the angiography-guided PCI group. Inverse probability of treatment weighting (IPTW) was performed using propensity scores with a logistic regression model that included baseline covariates. The balance between groups after weighting was evaluated using absolute standardised mean differences, with values ≤0.1 considered negligible. Hazard ratios (HRs) were calculated using the angiography-guided PCI group as a reference. Statistical significance was set at p<0.05.
Subgroup analyses of the primary outcome were conducted based on age, sex, diabetes mellitus, peripheral vascular disease, and acute myocardial infarction, using multivariable Cox proportional hazards models.
Sensitivity analyses were conducted to assess the effects of the alternative statistical methods and study definitions. First, a multivariable Cox regression analysis was performed, based on the same covariates, to conduct a complementary analysis comparing the groups. Second, propensity score matching was performed using a multivariable Cox proportional hazards model. A calliper width of 0.1 times the standard deviation of the logit of the propensity score and 1:1 nearest-neighbour matching without replacement were used. Third, the analysis was focused on patients with drug-eluting stent (DES) implantation to reflect the inclusion criteria of previous RCTs, and IPTW was performed for the primary and secondary outcomes based on the same covariates. Fourth, to assess effects by index year, the cumulative incidence of ACS recurrence and the absolute risk difference were reported for 2014-2018, with complete 3-year follow-up. Finally, we performed IPTW using hip fracture as a negative control outcome. The data were analysed using Stata version 17 (StataCorp).
Results
Study population and baseline characteristics
A total of 492,303 patients with ACS who underwent their first PCI procedure between April 2014 and March 2021 were identified (Figure 1, Central illustration). The annual changes in the utilisation rate of intravascular imaging are shown in Figure 2. During the study period, OCT-guided PCI increased from 4.7% to 6.9%, and IVUS-guided PCI increased from 77.0% to 87.9%, while angiography-guided PCI without the use of intravascular imaging decreased from 17.0% to 4.8%. From the total number of screened patients, 355,811 met the eligibility criteria and were included in the study. Among the participants, 85,960 (24.2%) were aged 60-69 years, 111,396 (31.3%) were aged 70-79 years, and 86,866 (24.4%) were female. The patients were divided into the imaging-guided PCI group (OCT-guided: 22,748 patients; IVUS-guided: 297,944 patients) and the angiography-guided PCI group (32,044 patients). The baseline characteristics according to the groups are shown in Table 1 and Supplementary Table 2. The imaging-guided PCI group had a higher proportion of patients with unstable angina, DES implantation, and procedures in more recent fiscal years, while the angiography-guided PCI group had higher proportions of elderly patients, females, comorbidities, a past medical history, acute myocardial infarction, and procedures performed in earlier fiscal years.
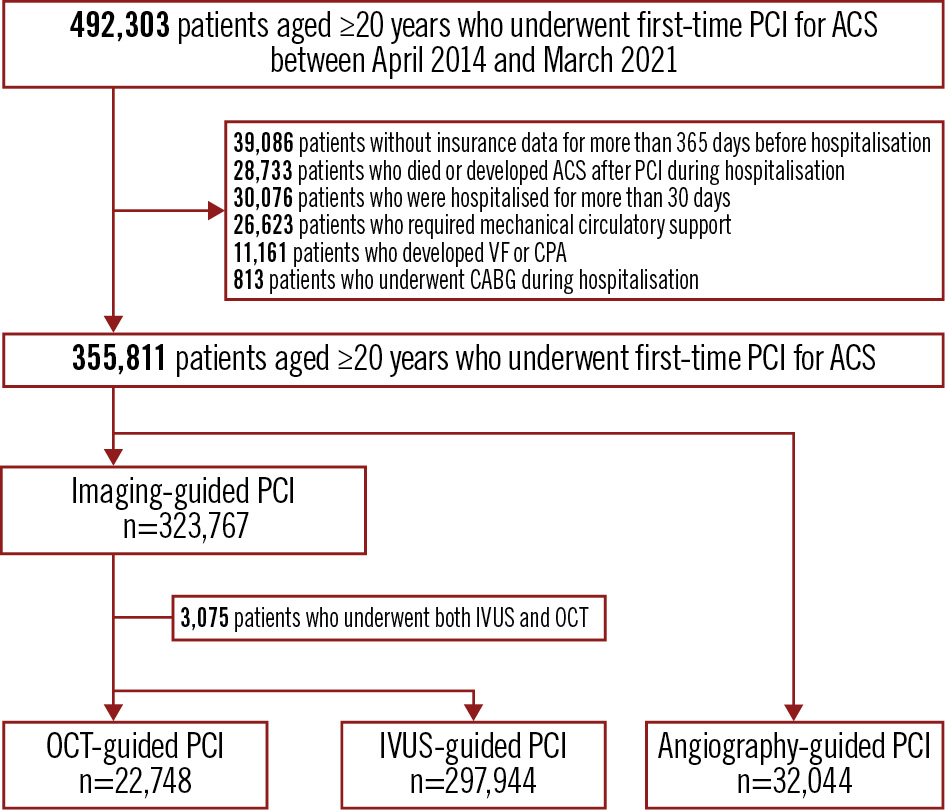
Figure 1. Flowchart of patient selection. A total of 492,303 patients who underwent first-time PCI for ACS and were discharged alive were screened. ACS: acute coronary syndrome; CABG: coronary artery bypass grafting; CPA: cardiopulmonary arrest; IVUS: intravascular ultrasound; OCT: optical coherence tomography; PCI: percutaneous coronary intervention; VF: ventricular fibrillation
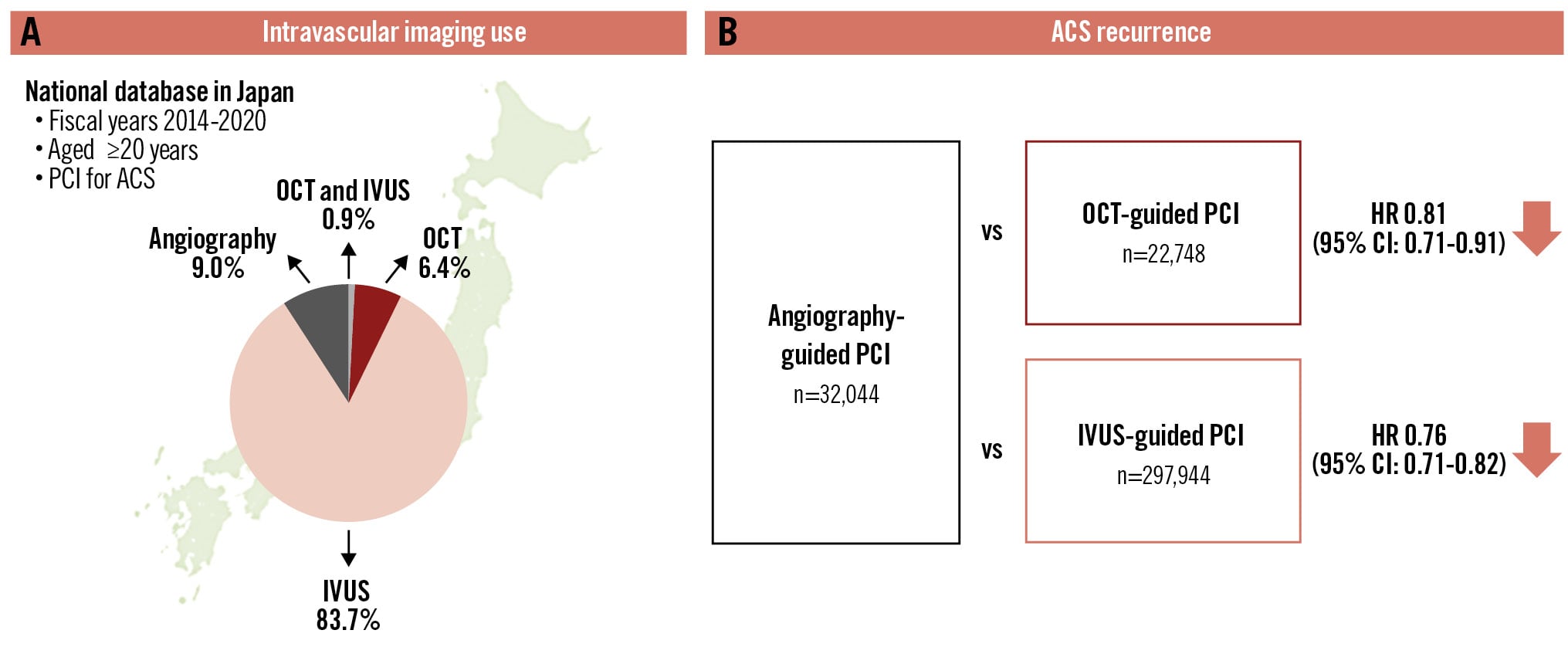
Central illustration. Intravascular imaging-guided percutaneous coronary intervention in patients with acute coronary syndrome. A) Intravascular imaging use. B) Recurrence of ACS. ACS: acute coronary syndrome; CI: confidence interval; HR: hazard ratio; IVUS: intravascular ultrasound; OCT: optical coherence tomography; PCI: percutaneous coronary intervention
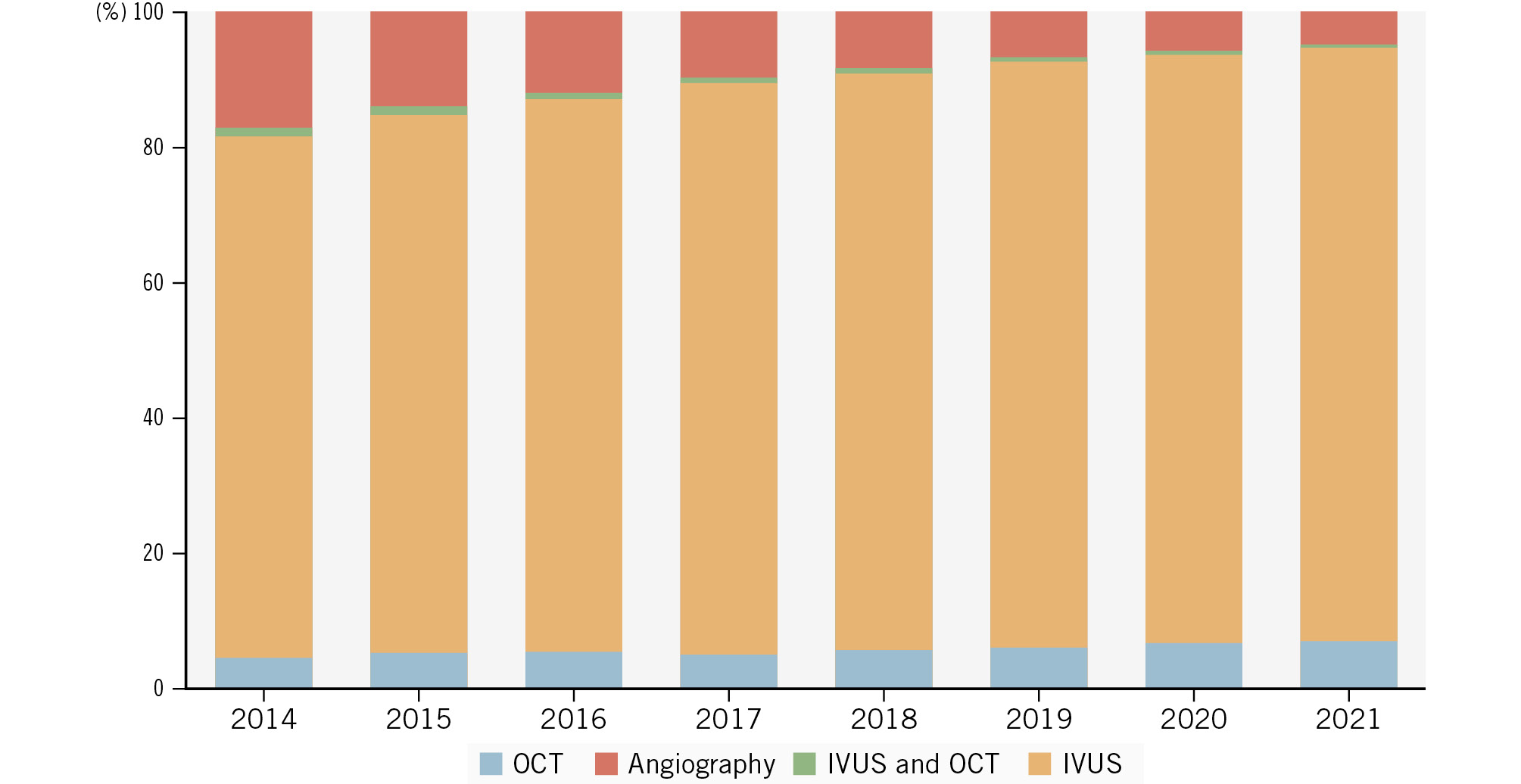
Figure 2. Annual changes in the utilisation rate of intravascular imaging in patients with ACS. ACS: acute coronary syndrome; IVUS: intravascular ultrasound; OCT: optical coherence tomography
Table 1. Baseline patient characteristics
| Angiography-guided PCI n=32,044 | OCT-guided PCI n=22,748 | IVUS-guided PCI n=297,944 | |
|---|---|---|---|
| Age category | |||
| <60 years | 5,900 (18.4) | 5,294 (23.3) | 62,707 (21.0) |
| 60-69 years | 7,453 (23.3) | 6,031 (26.5) | 71,655 (24.0) |
| 70-79 years | 9,911 (30.9) | 7,166 (31.5) | 93,442 (31.4) |
| ≥80 years | 8,780 (27.4) | 4,257 (18.7) | 70,140 (23.5) |
| Female | 9,094 (28.4) | 5,258 (23.1) | 71,783 (24.1) |
| Comorbidities/past medical history | |||
| Hypertension | 22,028 (68.7) | 14,611 (64.2) | 195,230 (65.5) |
| Diabetes mellitus | 7,996 (25.0) | 5,395 (23.7) | 73,631 (24.7) |
| Dyslipidaemia | 17,137 (53.5) | 12,606 (55.4) | 157,391 (52.8) |
| Atrial fibrillation | 1,421 (4.4) | 760 (3.3) | 11,182 (3.8) |
| Peripheral vascular disease | 4,730 (14.8) | 3,111 (13.7) | 37,915 (12.7) |
| Chronic kidney disease | 2,430 (7.6) | 1,368 (6.0) | 21,761 (7.3) |
| Medications during hospitalisation | |||
| Aspirin | 29,595 (92.4) | 21,408 (94.1) | 284,092 (95.4) |
| P2Y12 inhibitors | 29,688 (92.7) | 22,036 (96.9) | 289,687 (97.2) |
| OAC | 3,584 (11.2) | 1,837 (8.1) | 28,376 (9.5) |
| Statin | 23,908 (74.6) | 19,823 (87.1) | 255,617 (85.8) |
| Clinical presentation | |||
| Acute myocardial infarction | 22,255 (69.5) | 14,348 (63.1) | 201,320 (67.6) |
| Unstable angina | 9,789 (30.5) | 8,400 (36.9) | 96,624 (32.4) |
| Procedure during PCI | |||
| DES | 23,349 (72.9) | 19,993 (87.6) | 268,314 (90.1) |
| DCB | 2,085 (6.5) | 2,660 (11.7) | 22,229 (7.5) |
| Adjunctive atherectomy* | 515 (1.6) | 699 (3.1) | 6,864 (2.3) |
| Data are presented as n (%). *Adjunctive atherectomy refers to rotational atherectomy and orbital atherectomy. DCB: drug-coated balloon; DES: drug-eluting stent; IVUS: intravascular ultrasound; OAC: oral anticoagulant; OCT: optical coherence tomography; PCI: percutaneous coronary intervention | |||
Outcomes of imaging- versus angiography-guided PCI
The median follow-up period after PCI was 1,113 days (interquartile range 489-1,841 days). Crude event rates for the primary and secondary outcomes during the 3-year follow-up are shown in Supplementary Table 3 and Supplementary Figure 2. We performed an analysis focusing on the comparison between the imaging-guided PCI group and the angiography-guided PCI group. After weighting, all baseline covariates had an absolute standardised mean difference below 0.1, indicating a good balance between groups (Supplementary Figure 3). Weighted clinical outcomes for the primary and secondary outcomes over the 3-year follow-up are presented in Supplementary Table 4 and Supplementary Figure 4. The incidence of the primary outcome was lower in the imaging-guided PCI group (HR 0.76, 95% confidence interval [CI]: 0.70-0.82; p<0.001), and the incidence of the secondary outcome was also lower in the imaging-guided PCI group (HR 0.85, 95% CI: 0.82-0.89; p<0.001).
Outcomes of OCT- or IVUS-guided PCI versus angiography-guided PCI
We performed a subsequent analysis focusing on the imaging-guided (OCT or IVUS) PCI group. Crude event rates for the primary and secondary outcomes during the 3-year follow-up are shown in Supplementary Table 3 and Supplementary Figure 5. Crude event rates for the primary outcome were 586 patients (1.22 per 100 person-years) in OCT-guided PCI, 7,101 patients (1.11 per 100 person-years) in IVUS-guided PCI, and 1,121 patients (1.47 per 100 person-years) in angiography-guided PCI. Adjusted clinical outcomes for the primary and secondary outcomes over the 3-year follow-up are presented in Table 2, Supplementary Table 5, Figure 3, Supplementary Figure 6, and Supplementary Figure 7. The incidence of the primary outcome was lower in the OCT-guided PCI group (HR 0.81, 95% CI: 0.71-0.91; p<0.001) and in the IVUS-guided PCI group (HR 0.76, 95% CI: 0.71-0.82; p<0.001), and the incidence of the secondary outcome was also lower in the OCT-guided PCI group (HR 0.80, 95% CI: 0.74-0.87; p<0.001) and in the IVUS-guided PCI group (HR 0.86, 95% CI: 0.83-0.90; p<0.001) compared with the angiography-guided PCI group.
Table 2. Primary outcome (at 3 years) of OCT- and IVUS-guided PCI versus angiography-guided PCI.
| OCT- and IVUS-guided PCI | Angiography-guided PCI | HR (95% CI) | p-value | |||
|---|---|---|---|---|---|---|
| Total events | Incidence rate/100 person-years (95% CI) | Total events | Incidence rate/100 person-years (95% CI) | |||
| OCT-guided PCI vs angiography-guided PCI | ||||||
| IPTW | 647/22,731 | 1.25 (1.12-1.38) | 1,117/32,199 | 1.54 (1.44-1.64) | 0.81 (0.71-0.91) | <0.001 |
| Multivariable Cox | 586/22,748 | 1.22 (1.13-1.32) | 1,121/32,044 | 1.47 (1.39-1.56) | 0.75 (0.68-0.84) | <0.001 |
| PS matching | 495/17,857 | 1.20 (1.10-1.31) | 619/17,857 | 1.54 (1.42-1.66) | 0.78 (0.69-0.88) | <0.001 |
| IVUS-guided PCI versus angiography-guided PCI | ||||||
| IPTW | 7,247/297,979 | 1.12 (1.09-1.14) | 1,016/32,053 | 1.46 (1.36-1.57) | 0.76 (0.71-0.82) | <0.001 |
| Multivariable Cox | 7,101/297,944 | 1.11 (1.08-1.13) | 1,121/32,044 | 1.47 (1.39-1.56) | 0.78 (0.73-0.83) | <0.001 |
| PS matching | 925/31,951 | 1.20 (1.13-1.28) | 1,115/31,951 | 1.47 (1.38-1.55) | 0.82 (0.75-0.89) | <0.001 |
| Data are presented as n/N. Models were adjusted using the baseline covariates, including age, sex, comorbidities, prescribed medications, PCI procedure, and fiscal year. CI: confidence interval; HR: hazard ratio; IPTW: inverse probability of treatment weighting; IVUS: intravascular ultrasound; OCT: optical coherence tomography; PCI: percutaneous coronary intervention; PS: propensity score | ||||||
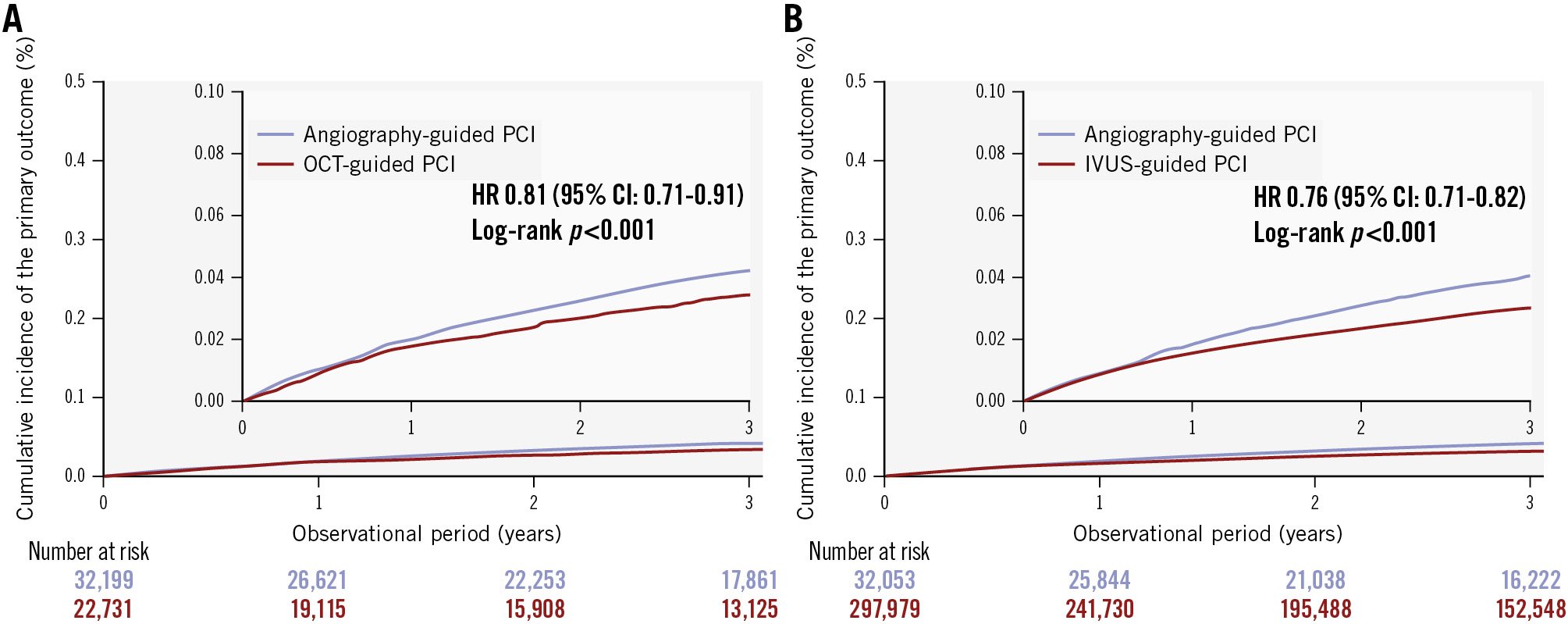
Figure 3. Kaplan-Meier curves for the primary outcome (ACS recurrence) after IPTW. A) OCT-guided PCI versus angiography-guided PCI; (B) IVUS-guided PCI versus angiography-guided PCI. ACS: acute coronary syndrome; CI: confidence interval; HR: hazard ratio; IPTW: inverse probability of treatment weighting; IVUS: intravascular ultrasound; OCT: optical coherence tomography; PCI: percutaneous coronary intervention
Subgroup analysis
The results of the subgroup analysis are shown in Supplementary Figure 8. No interactions were observed between subgroups based on age, sex, or diabetes mellitus. On the other hand, an interaction was found for peripheral vascular disease (p=0.013) and acute myocardial infarction (p<0.001), showing a lower incidence of ACS recurrence in the imaging-guided PCI group.
Sensitivity analysis
The sensitivity analysis results are shown in Supplementary Table 4 - Supplementary Table 5 - Supplementary Table 6 - Supplementary Table 7 - Supplementary Table 8 - Supplementary Table 9 - Supplementary Table 10, Supplementary Figure 4, and Supplementary Figure 9 - Supplementary Figure 10 - Supplementary Figure 11 - Supplementary Figure 12 - Supplementary Figure 13. When the inclusion criteria were limited to patients without heart failure (HF) or those who underwent DES implantation, ACS recurrence was lower in the imaging-guided PCI group compared with the angiography-guided PCI group. The cumulative incidence of ACS recurrence and the absolute risk difference are presented in Supplementary Table 11. There was no difference between the groups for hip fracture (used as a negative control) (Supplementary Table 12, Supplementary Figure 14).
Discussion
This study investigated the association between imaging-guided PCI and clinical outcomes in patients with ACS, compared with angiography-guided PCI, using the NDB, which covers almost all patients in Japan. Our findings indicate that intravascular imaging was used in over 90% of cases, and both OCT- and IVUS-guided PCI were associated with a lower recurrence of ACS compared with angiography-guided PCI. Sensitivity analysis supported the robustness of these findings.
The proportion of imaging-guided PCI increased annually, while that of angiography-guided PCI without the use of intravascular imaging declined. This trend aligns with a global increase in the use of intravascular imaging reported in other countries67. Among the intravascular imaging modalities, IVUS was used more frequently than OCT in ACS patients. The proportion of OCT-guided PCI gradually increased to 6.9%. This lower use of OCT may be due to the requirements both for contrast agents in patients with chronic kidney disease and for technical expertise in OCT-guided PCI524252627. In Europe and America, the use of intravascular imaging during PCI has been reported to be low2829. The findings of this study and the IVUS-ACS trial align with current guideline recommendations12.
Imaging-guided PCI for ACS was associated with a lower recurrence of ACS. A previous RCT demonstrated a reduction in target vessel failure at the 1-year follow-up in IVUS-guided PCI compared with angiography-guided PCI in patients with ACS1017. Our study, including patients in real-world settings with a 3-year follow-up, demonstrated consistent results. These results imply that imaging-guided PCI for ACS could be useful in generalisable populations, and they may promote imaging-guided PCI in clinical practice.
OCT-guided PCI was associated with fewer negative outcomes compared with angiography-guided PCI in patients with ACS. OCT provides higher resolution than IVUS, allowing for a more detailed assessment of culprit lesions and optimal stent implantation. One RCT has reported that OCT-guided PCI resulted in a lower incidence of target vessel failure compared with angiography-guided PCI in patients with complex coronary artery lesions14. There have been few large-scale studies that have investigated the impact of OCT-guided PCI on long-term outcomes303132333435; our results suggest that OCT-guided PCI may be an important option in patients with ACS.
This study was the largest to date on the use of imaging-guided PCI, including both OCT and IVUS, for ACS patients in a real-world setting. However, RCTs in patients with ACS are still lacking. Further RCTs are warranted to establish evidence for the use of imaging-guided PCI, including OCT-guided PCI, in patients with ACS.
Limitations
This study has several limitations due to its observational design. Although we adjusted for comorbidities such as dialysis and chronic kidney disease, the NDB does not include detailed data on factors such as renal function and activities of daily living, which may have led to residual confounding. Second, this study does not include detailed intravascular imaging findings. Third, since this study is based on the Japanese population, its findings may not be directly applicable to other geographic regions. Finally, we did not evaluate costs or cost-effectiveness, as our claims data lacked itemised device prices.
Conclusions
In this nationwide study, both OCT- and IVUS-guided PCI were associated with a reduction in ACS recurrence. These findings highlight the potential benefits of imaging-guided PCI in ACS management. In patients with ACS, increased utilisation of intravascular imaging may contribute to a reduction in ACS recurrence.
Impact on daily practice
Intravascular imaging (optical coherence tomography [OCT] and intravascular ultrasound [IVUS]) during percutaneous coronary intervention (PCI) provides detailed lesion assessment and stent optimisation, and, in this nationwide analysis, was associated with lower acute coronary syndrome (ACS) recurrence compared with angiography guidance. The integration of imaging guidance into PCI practice can improve procedural safety by identifying stent malapposition, underexpansion, and periprocedural complications, thereby enhancing long-term clinical outcomes in patients with ACS. Further randomised trials specifically targeting ACS populations are needed to validate the long-term benefit of OCT- and IVUS-guided PCI and to inform global guideline recommendations.
Funding
This work was supported by the Japan Society for the Promotion of Science KAKENHI grant 24K19047 and the Asahipen Hikari Foundation grant JP25APH621 for the publication of this article.Conflict of interest statemenK. Kanaoka has received speaker honoraria from Eli Lilly Japan and Otsuka Pharmaceutical Co.; and has received consultation fees from Johnson & Johnson. The other authors have no conflicts of interest to declare.
Supplementary data
To read the full content of this article, please download the PDF.

