Cory:
Unlock Your AI Assistant Now!
Abstract
Background: Endovascular therapy is a cornerstone for chronic limb-threatening ischaemia (CLTI), yet the optimal device strategy remains uncertain.
Aims: Our objective was to compare the clinical and economic outcomes between plain balloon angioplasty±bare metal stents (PBA±BMS), drug-coated balloons (DCBs)±BMS, and drug-eluting stents (DES) in a national real-world CLTI cohort.
Methods: Medicare beneficiaries aged ≥66 years who underwent femoropopliteal revascularisation for CLTI between 2016 and 2023 were included. Patients were grouped by index device. Outcomes included a composite of all-cause mortality or major amputation, as well as major adverse limb events (MALE) and reintervention. Patients were followed from the index procedure until death, loss to follow-up, or the end of the study period. Time-to-event and cost outcomes were analysed using multivariable Cox and gamma regression models, respectively.
Results: Among 108,304 CLTI patients, 52.5% received PBA±BMS, 30.7% DCBs, and 16.8% DES. At 2 years, the composite outcome occurred in 50.54% (PBA±BMS), 43.08% (DCB±BMS), and 43.71% (DES); at 5 years, it occurred in 75.69%, 71.19%, and 71.71%, respectively. Compared with PBA±BMS, DCB±BMS (hazard ratio [HR] 0.92, 95% confidence interval [CI]: 0.90-0.93) and DES (HR 0.93, 95% CI: 0.92-0.95) were associated with a lower risk of the composite outcome. DCBs were associated with reduced major amputation (HR 0.87, 95% CI: 0.84-0.91), mortality (HR 0.93, 95% CI: 0.91-0.94), MALE (HR 0.96, 95% CI: 0.94-0.98), and reintervention (HR 0.97, 95% CI: 0.96-0.99) compared with PBA±BMS. The proportion of BMS use was 10.1% in the PBA±BMS group and 3.1% in the DCB±BMS group.
Conclusions: In this national CLTI cohort, drug-coated devices were associated with reduced amputation and mortality. Data from this study suggest that DCBs may offer consistent benefit without increased costs.
Chronic limb-threatening ischaemia (CLTI) is defined by ischaemic rest pain, non-healing ulcers, or gangrene and is associated with high morbidity and mortality, with amputation or death occurring in up to 25% of patients within 1 year if left untreated123. Prompt revascularisation is the cornerstone of therapy and is essential to improve limb salvage and survival45.
Over the past decade, endovascular therapy has emerged as a first-line strategy complementing open surgery in patients with CLTI due to its less invasive nature, rapid recovery, and evolving technological innovation367. Although drug-eluting technology has become the most frequently used strategy for the femoropopliteal (FP) segment, its efficacy and safety have been incompletely studied in the CLTI population because of limited representation in device approval trials8. In response to this lack of data, the BASIL-3 randomised trial was conducted in the United Kingdom to evaluate the impact of drug-coated devices on CLTI outcomes9. Despite its pragmatic design and clinical relevance, the trial had challenges with enrolment and, in a limited sample, did not identify differences between drug-coated and non-drug-coated devices for improving amputation-free survival.
Given its pragmatic design, BASIL-3 nonetheless provided important insights into contemporary endovascular management, although its external validity remains limited. Similarly, the BASIL-2 trial – focused on infrapopliteal disease – enrolled only a small and highly selected subset of patients, with limited generalisability to the broader CLTI population910. We aimed to assess whether the results of BASIL-3 were generalisable to a broader and more diverse CLTI population, characterised by older age; greater comorbidity burden, ethnic and socioeconomic diversity; and varied access to care111213. The objective of this observational cohort study was to emulate BASIL-3 in a national Medicare population and compare the safety, effectiveness, and costs of plain balloon angioplasty±bare metal stents (PBA±BMS), drug-coated balloons (DCBs)±BMS, and drug-eluting stents (DES) for FP revascularisation in patients with CLTI.
Methods
Study design and data sources
This retrospective observational cohort study was conducted using all complete Medicare Fee-For-Service (FFS) claims data covering the period from 1 January 2016 to 31 December 2023. Incomplete or partially adjudicated claims were excluded to ensure comprehensive procedural and outcome ascertainment. The data sources included the Master Beneficiary Summary File (MBSF) for demographic and enrolment data, Inpatient files for inpatient claims, and the Outpatient and Carrier files for institutional and physician services. Information on chronic comorbidities was obtained from the Chronic Condition Data Warehouse using validated algorithms. Procedures and devices were identified through a combination of ICD-10-CM, ICD-10-PCS, Current Procedural Terminology (CPT), and Healthcare Common Procedure Coding System (HCPCS) codes as detailed in Supplementary Table 1-Supplementary Table 2-Supplementary Table 3-Supplementary Table 4-Supplementary Table 5-Supplementary Table 6-Supplementary Table 7-Supplementary Table 8-Supplementary Table 9-Supplementary Table 10-Supplementary Table 11-Supplementary Table 12-Supplementary Table 13-Supplementary Table 1414. To ensure consistent longitudinal ascertainment, all codes were selected based on their validity across the entire study period (2016-2023). When coding changes occurred (e.g., code retirements, updates, or replacements), older codes were mapped to their most accurate contemporary equivalents through expert clinical review, and crosswalks were provided by Centers for Medicare & Medicaid Services (CMS) and American Hospital Association coding guidelines, allowing harmonisation across all years. The study protocol was approved by the Institutional Review Board of Beth Israel Deaconess Medical Center, with a waiver of informed consent granted given the retrospective design. This study followed the STROBE guidelines for reporting observational cohort studies15.
Study population
All Medicare beneficiaries aged 66 years and older who underwent percutaneous endovascular revascularisation for CLTI involving the FP segment during the study period were included. CLTI was defined by the presence of ischaemic rest pain, tissue loss, or gangrene as captured through administrative claims (Supplementary Table 1-Supplementary Table 2-Supplementary Table 3). Patients with a procedure date between 2016 and 2023 were included. For each patient, continuous enrolment was verified in Medicare FFS during the 12 months preceding their index procedure using the MBSF, which was available from 2015 onwards. In order to allow for consistent baseline characterisation, only patients with at least 12 months of continuous enrolment in Medicare FFS before the index procedure were included. The first qualifying procedure within the study period was considered the index intervention, and all subsequent procedures were considered outcome events. Patients were excluded if they had a prior major lower extremity amputation or if critical procedural data, including device type, were missing or unidentifiable in their claims. Because claims submitted by privately-owned facilities, such as office-based laboratories and ambulatory surgical centres, do not report the device information needed to identify drug-coated technologies, the procedures performed in these settings were excluded from the primary analysis to ensure accurate classification of treatment strategy, as has been the approach in prior work16. Patient selection is summarised in Supplementary Figure 1.
Device classification
Patients were categorised into three mutually exclusive treatment groups based on the devices used during the index procedure to emulate the BASIL-3 trial. The PBA±BMS group included patients treated with PBA, with or without the use of BMS. The DCB±BMS group included patients who received DCB angioplasty, with or without adjunctive BMS implantation. The DES group included those who received drug-eluting stents, regardless of whether DCBs or BMS were additionally used. In cases where multiple device types were coded within the same intervention, a hierarchical classification was applied, with DES prioritised over DCBs and DCBs prioritised over PBA±BMS, following the same logic used in prior Medicare-based device evaluations11.
Covariates
Baseline covariates included demographic characteristics (age, sex, and race/ethnicity), markers of socioeconomic vulnerability (dual Medicare and Medicaid eligibility status, defined as concurrent enrolment in both programmes and serving as a proxy for low income and socioeconomic disadvantage in the US healthcare system; residential community-level economic distress based on the Distressed Communities Index; and location in a rural or urban area), and comorbid conditions (such as diabetes; tobacco use; chronic kidney disease; ischaemic heart disease; congestive heart failure; myocardial infarction; hypertension; hyperlipidaemia; obesity; Alzheimer’s disease, related disorders or senile dementia; cancer; chronic obstructive pulmonary disease; and stroke/transient ischaemic attack). CLTI severity was characterised using claims-based indicators of rest pain, ulceration, and gangrene. The care setting of the primary procedure was categorised as either hospital inpatient or hospital outpatient. Additional covariates included geographic region, calendar year of treatment, and the arterial segment treated (femoropopliteal alone, femoropopliteal with tibial, or multilevel interventions), concomitant use of atherectomy, annual institutional endovascular revascularisation volume, academic status, and hospital bed size. The standardised mean differences of baseline covariates between groups before and after matching are shown in Supplementary Figure 2 and Supplementary Figure 3.
Outcomes
The primary outcome of interest was defined as the composite of all-cause mortality or major amputation of the lower extremity. Major amputation was defined as an above-the-ankle amputation (Supplementary Table 15-Supplementary Table 16-Supplementary Table 17-Supplementary Table 18-Supplementary Table 19-Supplementary Table 20-Supplementary Table 21). Secondary outcomes included all-cause mortality, major amputation alone, major adverse limb events (MALE; defined as any repeat intervention or major amputation), major adverse cardiovascular events (MACE; including myocardial infarction, stroke, or death), and the need for repeat endovascular or surgical reintervention to either limb. Healthcare utilisation was assessed by index procedure length of stay and post-discharge emergency room visits and hospitalisations. Additionally, total healthcare costs were evaluated, including overall charges, Medicare payments, and provider payments, to assess the economic impact of each endovascular strategy.
Statistical analysis
Continuous variables are summarised using means with standard deviations or medians with interquartile ranges as appropriate, and categorical variables are presented as frequencies with corresponding percentages. Differences in baseline characteristics were evaluated using standardised mean differences (SMDs), with an SMD greater than 10% considered indicative of a meaningful imbalance17. Cumulative incidences of the primary outcome were estimated using Kaplan-Meier methods. For outcomes that did not include death, the cumulative incidence function was used to address the competing risk of death18. To account for patient clustering within hospitals, marginal Cox regression was used to compare outcomes between treatment groups, with adjustment for all the variables specified above19. Due to the long follow-up periods and high frequency of death in this population, incidences of events were reported at 2 years, 5 years, and longest follow-up.
We used Cox proportional hazards and Fine-Gray models for time-to-event outcomes, and Poisson and gamma regressions for healthcare utilisation and cost analyses, adjusting for predefined covariates (Supplementary Table 22). The proportional hazards assumption was tested and met for all Cox models.
Subgroup analyses were conducted by age (66-75 years, 76-85 years, >85 years), sex, race/ethnicity, community distress, geographic region, arterial segment treated (femoropopliteal alone, femoropopliteal and tibial, or multilevel), and rural versus urban location. To further assess the robustness of our findings, sensitivity analyses included inverse probability of treatment weighting and falsification endpoints (pneumonia and hip fracture)20.
Temporal trends in the use of each endovascular modality were evaluated across calendar years using linear regression models. The annual proportion of patients receiving PBA±BMS, DCB±BMS, or DES was calculated, and trend estimates were reported with corresponding 95% confidence intervals (CIs) and p-values.
Statistical analyses were conducted using SAS software, version 9.4 (SAS Institute). A 2-sided p-value below 0.05 was considered statistically significant. There were no missing data for key demographic, procedural, or outcome variables, as the CMS claims datasets used in this analysis provide complete records for the variables of interest. Although claims data may lack clinical granularity, all the variables required for inclusion and modelling were available for the entire cohort.
Results
Study population
Between 1 January 2016 and 31 December 2023, a total of 108,304 patients underwent FP endovascular revascularisation for CLTI and were included in the final analytic cohort. Among them, 56,875 (52.5%) patients received PBA±BMS, 33,276 (30.7%) were treated with DCB±BMS, and 18,153 (16.8%) received DES during their index procedure (Supplementary Table 23). The proportion of BMS use was 10.1% in the PBA±BMS group and 3.1% in the DCB±BMS group. The mean age was 77.76±7.97 years, and 46.3% of the cohort were females. Black patients represented 12.64% of the population, and 19.93% of all patients were dually enrolled in Medicare and Medicaid. Overall, 68.9% of patients presented with tissue loss, including ulceration (38.2%) or gangrene (30.68%). Comorbid conditions were highly prevalent, with 69.13% of patients having diabetes and 72.15% being diagnosed with chronic kidney disease. Compared with the PBA±BMS group, patients treated with DCB±BMS or DES were younger, more frequently treated in hospital outpatient settings, and more often managed in urban regions and high-volume centres. The distribution of baseline characteristics by treatment group is detailed in Table 1.
Table 1. Baseline characteristics of patients who underwent CLTI fempop endovascular revascularisation with laterality, stratified by modality category − primary analytic cohort.
| Subject characteristics | All patients (N=80,613) | PBA±BMS (N=48,955) | DCB±BMS (N=20,165) | DES (N=11,493) | SMD: DCB vs PBA±BMS | SMD: DES vs PBA±BMS | p-value |
|---|---|---|---|---|---|---|---|
| Age, years | 77.77±8.03 | 77.82±8.07 | 77.67±7.97 | 77.74±7.97 | –1.8 | –1.0 | 0.084 |
| Age, years | 77 [71-84] | 77 [71-84] | 77 [71-84] | 77 [71-84] | - | - | 0.084 |
| Female | 36,801 (45.65) | 22,534 (46.03) | 9,319 (46.21) | 4,948 (43.05) | 0.4 | –6.0 | <0.001 |
| Race/ethnicity | <0.001 | ||||||
| White | 63,985 (79.37) | 38,640 (78.93) | 16,005 (79.37) | 9,340 (81.27) | 1.1 | 5.9 | |
| Black | 10,710 (13.29) | 6,723 (13.73) | 2,599 (12.89) | 1,388 (12.08) | –2.5 | –4.9 | |
| Asian | 1,003 (1.24) | 611 (1.25) | 263 (1.30) | 129 (1.12) | 0.5 | –1.2 | |
| Other | 4,915 (6.10) | 2,981 (6.09) | 1,298 (6.44) | 636 (5.53) | 1.4 | –2.4 | |
| Dual enrolment | 16,498 (20.47) | 10,048 (20.52) | 4,204 (20.85) | 2,246 (19.54) | 0.8 | –2.5 | 0.019 |
| Distressed community | 14,422 (18.47) | 8,917 (18.82) | 3,611 (18.48) | 1,894 (16.97) | –0.9 | –4.8 | <0.001 |
| Rural locations | 3,450 (4.29) | 2,070 (4.23) | 929 (4.61) | 451 (3.93) | 1.8 | –1.5 | 0.010 |
| Region | <0.001 | ||||||
| Northeast | 13,726 (17.33) | 8,051 (16.75) | 3,233 (16.28) | 2,442 (21.71) | –1.3 | 12.6 | |
| Midwest | 11,224 (14.17) | 6,537 (13.60) | 3,147 (15.85) | 1,540 (13.69) | 6.3 | 0.3 | |
| South | 42,043 (53.09) | 26,129 (54.35) | 10,344 (52.08) | 5,570 (49.52) | –4.5 | –9.7 | |
| West | 12,192 (15.40) | 7,359 (15.31) | 3,136 (15.79) | 1,697 (15.09) | 1.3 | –0.6 | |
| Medical comorbidities | |||||||
| Acute myocardial infarction | 14,258 (17.69) | 8,550 (17.47) | 3,627 (17.99) | 2,081 (18.11) | 1.4 | 1.7 | 0.117 |
| Atrial fibrillation | 27,171 (33.71) | 16,656 (34.02) | 6,775 (33.60) | 3,740 (32.54) | –0.9 | –3.1 | 0.010 |
| Chronic kidney disease | 59,410 (73.70) | 35,814 (73.16) | 15,288 (75.81) | 8,308 (72.29) | 6.1 | –2.0 | <0.001 |
| Heart failure | 48,690 (60.40) | 29,528 (60.32) | 12,350 (61.24) | 6,812 (59.27) | 1.9 | –2.1 | 0.002 |
| Diabetes | 56,351 (69.90) | 33,854 (69.15) | 14,574 (72.27) | 7,923 (68.94) | 6.9 | –0.5 | <0.001 |
| Hyperlipidaemia | 72,954 (90.50) | 44,359 (90.61) | 18,302 (90.76) | 10,293 (89.56) | 0.5 | –3.5 | <0.001 |
| Hypertension | 77,285 (95.87) | 47,004 (96.01) | 19,342 (95.92) | 10,939 (95.18) | –0.5 | –4.1 | <0.001 |
| Ischaemic heart disease | 64,480 (79.99) | 39,242 (80.16) | 16,066 (79.67) | 9,172 (79.81) | –1.2 | –0.9 | 0.303 |
| Stroke/TIA | 24,826 (30.80) | 15,183 (31.01) | 6,176 (30.63) | 3,467 (30.17) | –0.8 | –1.8 | 0.174 |
| Obesity | 28,959 (35.92) | 17,622 (36.00) | 7,408 (36.74) | 3,929 (34.19) | 1.5 | –3.8 | <0.001 |
| Tobacco dependency | 26,685 (33.10) | 16,206 (33.10) | 6,335 (31.42) | 4,144 (36.06) | –3.6 | 6.2 | <0.001 |
| Atherectomy | 36,534 (45.40) | 23,333 (47.77) | 9,514 (47.20) | 3,687 (32.11) | –1.2 | –32.4 | <0.001 |
| Peripheral artery disease severity | <0.001 | ||||||
| Rest pain | 23,186 (28.76) | 15,164 (30.98) | 4,815 (23.88) | 3,207 (27.90) | –16.0 | –6.7 | |
| Ulceration | 27,834 (34.53) | 16,226 (33.14) | 7,652 (37.95) | 3,956 (34.42) | 10.0 | 2.7 | |
| Gangrene | 29,593 (36.71) | 17,565 (35.88) | 7,698 (38.18) | 4,330 (37.68) | 4.8 | 3.7 | |
| Arterial segment of intervention categories | <0.001 | ||||||
| Fempop alone | 36,702 (45.53) | 23,785 (48.59) | 7,873 (39.04) | 5,044 (43.89) | –19.3 | –9.4 | |
| Iliac and fempop | 9,568 (11.87) | 5,750 (11.75) | 2,271 (11.26) | 1,547 (13.46) | –1.5 | 5.2 | |
| Fempop and tibial | 31,808 (39.46) | 17,936 (36.64) | 9,408 (46.66) | 4,464 (38.84) | 20.4 | 4.5 | |
| All levels | 2,535 (3.14) | 1,484 (3.03) | 613 (3.04) | 438 (3.81) | 0.0 | 4.3 | |
| Place of service | <0.001 | ||||||
| Hospital-based inpatient | 60,272 (74.77) | 36,061 (73.66) | 15,170 (75.23) | 9,041 (78.67) | 3.6 | 11.8 | |
| Hospital-based outpatient | 20,341 (25.23) | 12,894 (26.34) | 4,995 (24.77) | 2,452 (21.33) | –3.6 | –11.8 | |
| Hospital characteristics (excluding ASCs/OBLs) | |||||||
| Teaching hospital | 53,507 (67.41) | 32,758 (67.92) | 12,912 (64.96) | 7,837 (69.56) | –6.3 | 3.5 | <0.001 |
| Bed size | <0.001 | ||||||
| 6 to 49 | 730 (0.92) | 485 (1.01) | 171 (0.86) | 74 (0.66) | –1.5 | –3.8 | |
| 50 to 99 | 3,000 (3.78) | 1,914 (3.97) | 721 (3.63) | 365 (3.24) | –1.8 | –3.9 | |
| 100 to 299 | 29,207 (36.80) | 17,349 (35.97) | 7,800 (39.24) | 4,058 (36.02) | 6.8 | 0.1 | |
| 300 to 499 | 23,512 (29.62) | 14,524 (30.11) | 5,600 (28.17) | 3,388 (30.07) | –4.3 | –0.1 | |
| ≥500 | 22,923 (28.88) | 13,958 (28.94) | 5,584 (28.09) | 3,381 (30.01) | –1.9 | 2.3 | |
| Endovascular revascularisation volume (annual, all centres) | 108.22±82.78 | 105.65±81.08 | 111.89±83.87 | 112.69±87.46 | 7.6 | 8.3 | <0.001 |
| Surgical revascularisation volume (annual, all centres) | 18.09±15.99 | 18.30±16.12 | 17.56±15.71 | 18.11±15.87 | –4.6 | –1.1 | <0.001 |
| Length of stay of index procedure, days (all centres) | 6.96±8.64 | 7.03±8.80 | 6.78±8.51 | 6.95±8.11 | –2.8 | –0.9 | 0.003 |
| All values are n (%), median [interquartile range], or mean±standard deviation. ASC: ambulatory surgery centre; BMS: bare metal stent; CLTI: chronic limb-threatening ischaemia; DCB: drug-coated balloon; DES: drug-eluting stent; fempop: femoropopliteal; OBL: office-based laboratory; PBA: plain balloon angioplasty; SMD: standardised mean difference; TIA: transient ischaemic attack | |||||||
Trends in device utilisation for CLTI during the study period
From 2016 to 2023, the relative use of DES increased (+0.17%, 95% CI: 0.07-0.27%; p=0.001), while the use of DCB±BMS decreased slightly (–0.19%, 95% CI: –0.71 to 0.33%; p=0.46), and that of PBA±BMS remained stable (+0.02%, 95% CI: –0.53 to 0.57%; p=0.95) (Figure 1).
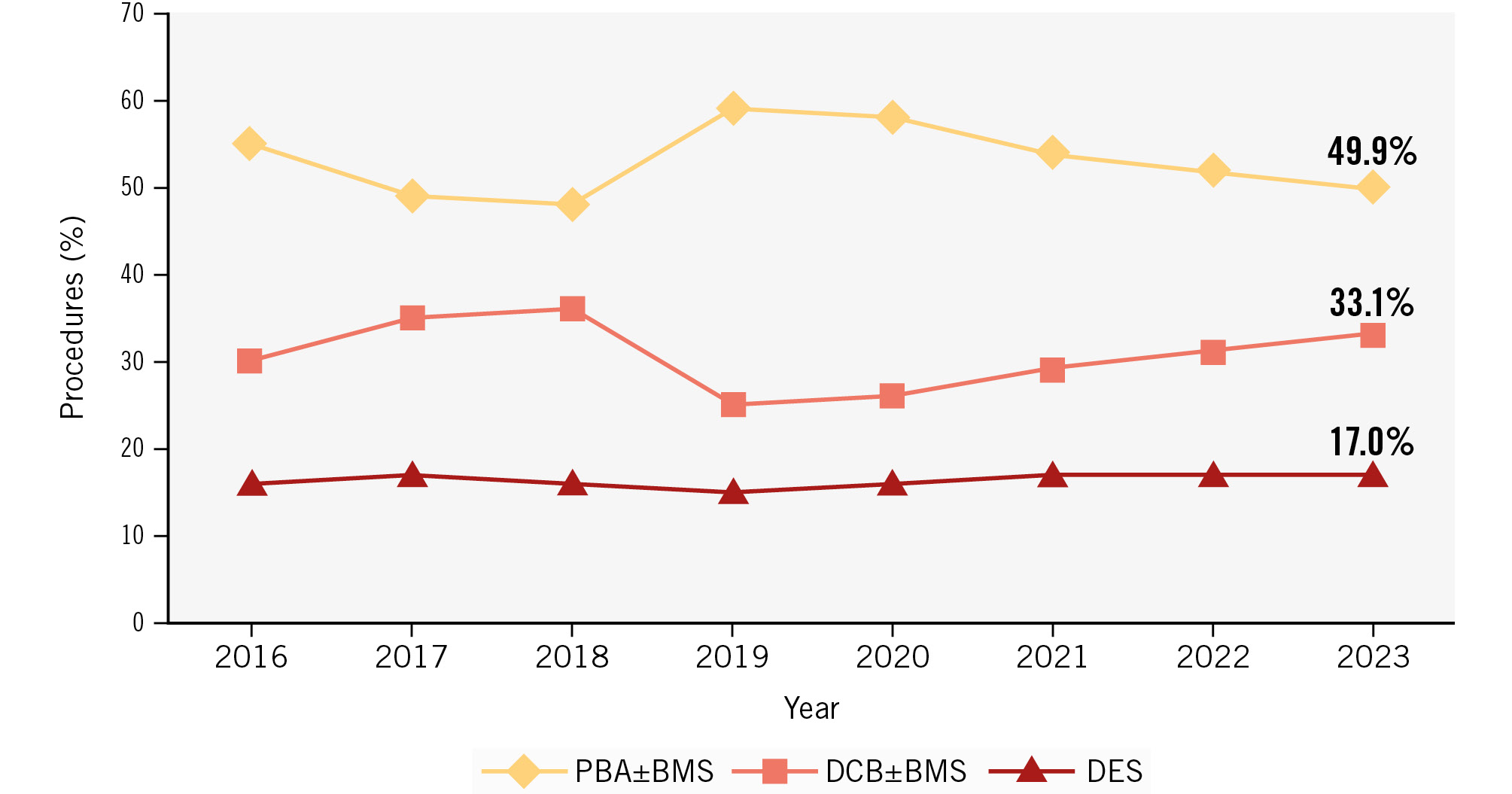
Figure 1. Revascularisation trends during the study period by modality. BMS: bare metal stent; DCB: drug-coated balloon; DES: drug-eluting stent; PBA: plain balloon angioplasty
Primary outcome results
Over a median follow-up period of 5.11 years (interquartile range: 2.88-6.81 years) and a maximum of 9.01 years, the cumulative incidence of the primary outcome was consistently lower in patients treated with DCB±BMS or DES compared with those treated with PBA±BMS.
At 2 years, the cumulative incidence was 50.54% (95% CI: 50.12-50.95) in the PBA±BMS group, 43.08% (95% CI: 42.54-43.62) in the DCB±BMS group, and 43.71% (95% CI: 42.97-44.44) in the DES group (p<0.001). The corresponding adjusted hazard ratios (HRs) were 0.88 (95% CI: 0.86-0.89; p<0.0001) for DCB±BMS, and 0.90 (95% CI: 0.88-0.93; p<0.0001) for DES.
At 5 years, the cumulative incidence reached 75.69% (95% CI: 75.28-76.09) in the PBA±BMS group, 71.19% (95% CI: 70.62-71.75) in the DCB±BMS group, and 71.71% (95% CI: 70.93-72.47) in the DES group, with adjusted HRs of 0.91 (95% CI: 0.90-0.93; p<0.0001) for DCB±BMS and 0.93 (95% CI: 0.91-0.95; p<0.0001) for DES (Figure 2).
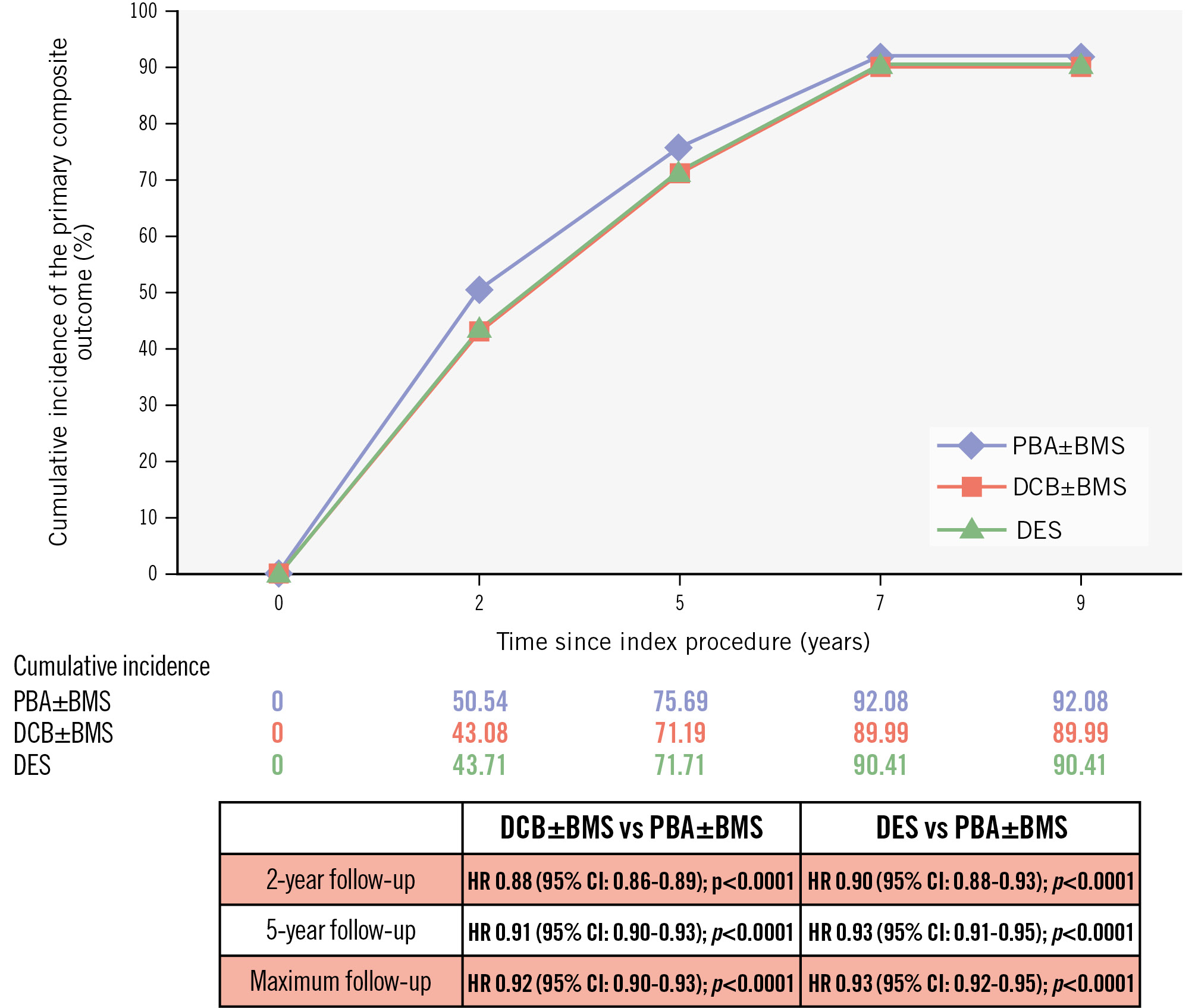
Figure 2. Cumulative incidence of the primary composite outcome: major amputation or all-cause death. BMS: bare metal stent; CI: confidence interval; DCB: drug-coated balloon; DES: drug-eluting stent; HR: hazard ratio; PBA: plain balloon angioplasty
Secondary outcomes results
Among the secondary endpoints, the composite endpoint occurred in 91.04% of the PBA±BMS group, 88.64% of the DCB±BMS group, and 89.15% of the DES group. Major amputation occurred in 14.85%, 12.82%, and 12.99% of patients, respectively. MALE rates were 48.19%, 50.29% and 50.80%, respectively. In a prespecified sensitivity analysis restricted to 80,613 patients with baseline procedural modifiers available (enabling laterality-specific event classification), the findings were consistent with those from the primary cohort. In this subgroup, ipsilateral major amputation occurred in 14.8% of PBA±BMS patients, 12.8% of DCB±BMS patients, and 13.0% of DES patients. Adjusted subdistribution HRs for ipsilateral major amputation were 0.89 (95% CI: 0.85-0.94; p<0.0001) for DCB±BMS and 0.93 (95% CI: 0.88-0.99; p=0.0292) for DES, compared with PBA±BMS (Supplementary Table 24).
After adjustment, DCB±BMS versus PBA±BMS was associated with lower risks of major amputation (HR 0.87, 95% CI: 0.84-0.91; p<0.0001), all-cause mortality (HR 0.93, 95% CI: 0.91-0.94; p<0.0001), MALE (HR 0.96, 95% CI: 0.94-0.98; p<0.0001), and any repeat revascularisation (HR 0.97, 95% CI: 0.96-0.99; p=0.0103). DES versus PBA±BMS was similarly associated with lower adjusted risks of major amputation (HR 0.90, 95% CI: 0.86-0.94; p<0.0001) and all-cause mortality (HR 0.94, 95% CI: 0.92-0.96; p<0.0001) but not MALE (HR 1.00, 95% CI: 0.98-1.02; p=0.8596) or repeat revascularisation (HR 1.02, 95% CI: 1.00-1.05; p=0.054). The risk of MACE was similar across all groups. Finally, neither DCB±BMS nor DES was associated with a meaningful reduction in emergency department evaluation or readmission risk after revascularisation compared with PBA±BMS (Figure 3).
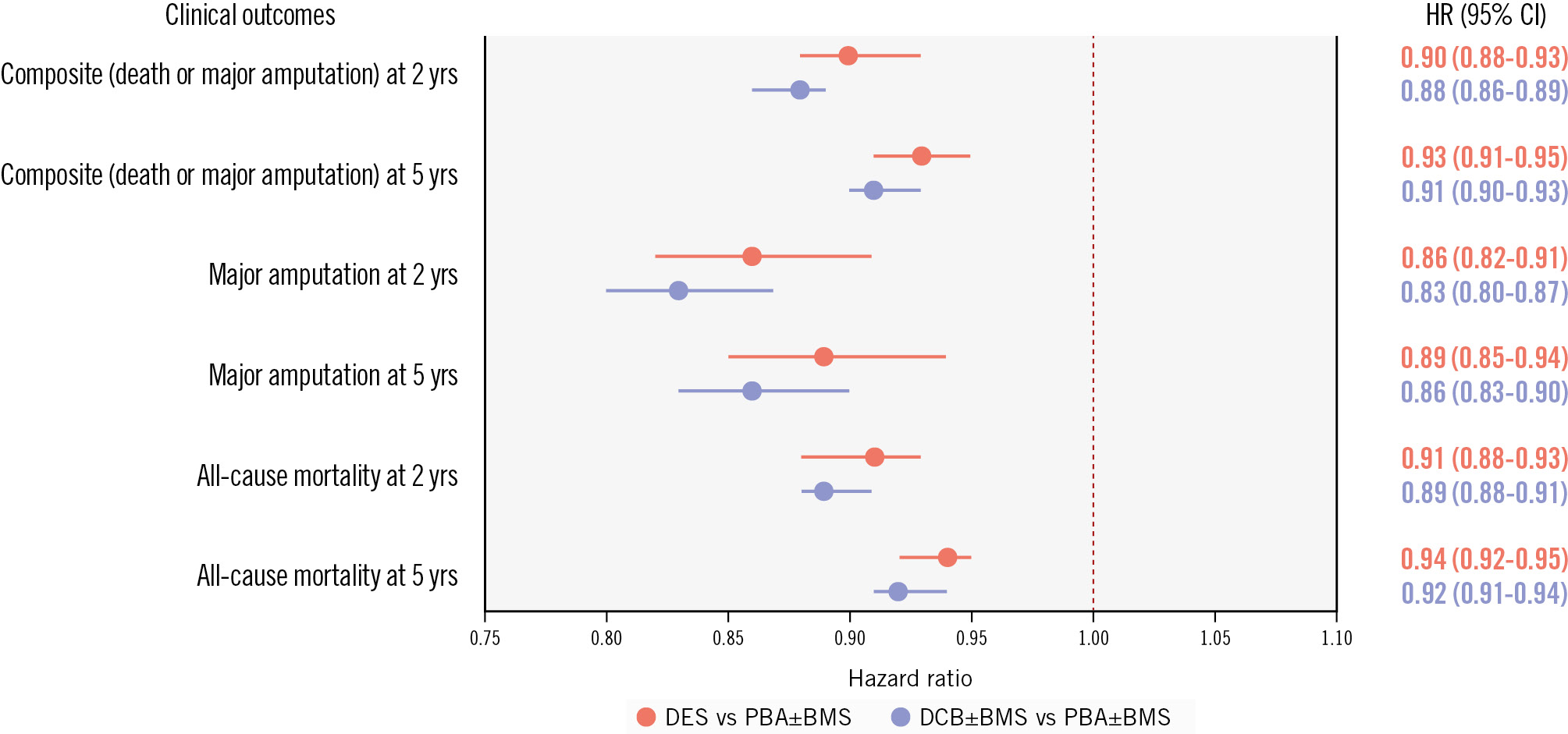
Figure 3. Risk of amputation or mortality with drug-coated versus uncoated endovascular devices at 2 and 5 years in CLTI. BMS: bare metal stent; CI: confidence interval; CLTI: chronic limb-threatening ischaemia; DCB: drug-coated balloon; DES: drug-eluting stent; HR: hazard ratio; PBA: plain balloon angioplasty
Multivariable analysis of predictors of the primary outcome
Multivariable Cox regression analyses revealed several patient-level factors independently associated with the primary outcome. Patients aged >85 years had a 30% higher risk of all-cause mortality or major amputation than those aged 76-85 years (adjusted HR 1.30, 95% CI: 1.28-1.33; p<0.0001), while patients aged 66-75 years had a 13% lower risk (vs 76-85 years: adjusted HR 0.87, 95% CI: 0.86-0.89; p<0.0001). Patients of female versus male sex had a lower risk of the primary outcome (adjusted HR 0.92, 95% CI: 0.91-0.93; p<0.0001), while Black versus White race was not significantly associated with the primary outcome (HR 1.00, 95% CI: 0.98-1.02; p=0.8586). Severe ischaemia was a major determinant of prognosis, with patients presenting with gangrene exhibiting nearly double the risk of amputation or death compared with those with rest pain (adjusted HR 1.83, 95% CI: 1.79-1.86; p<0.0001), and for those with ulcers, a 47% elevated risk of the primary outcome (adjusted HR 1.47, 95% CI: 1.44-1.49; p<0.0001). Chronic kidney disease, diabetes, tobacco dependence, and dual enrolment were also associated with worse outcomes (Figure 4).
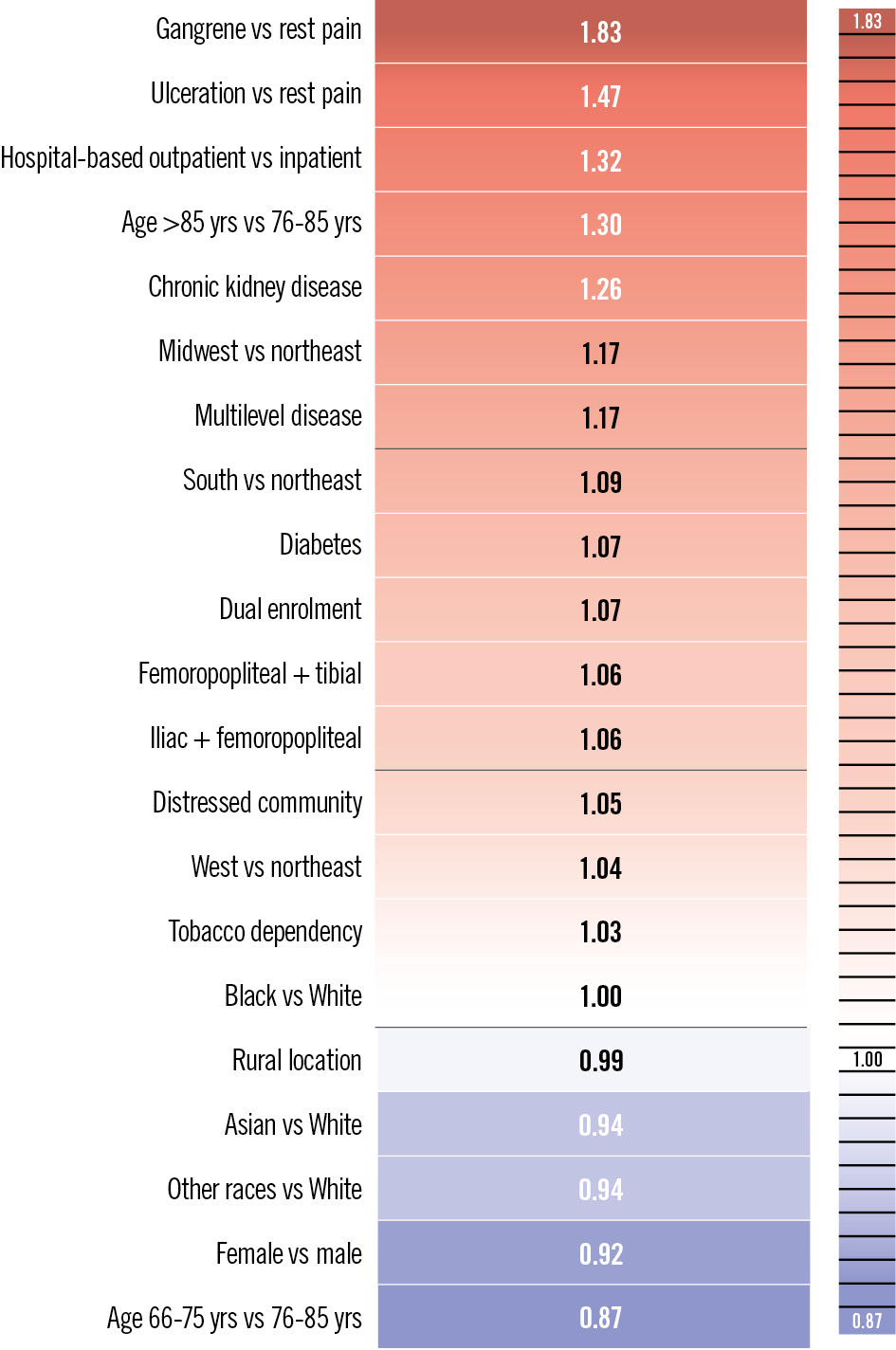
Figure 4. Adjusted hazard ratios for the primary composite outcome of all-cause mortality or major amputation. Hazard ratios were derived from multivariable regression models and adjusted for demographic, clinical, socioeconomic, and geographical factors. Values >1 indicate increased risk, while values <1 indicate decreased risk relative to the reference category for each covariate.
Sensitivity analysis
In sensitivity analyses, results were consistent across multiple approaches. When stratified by arterial segment, patients with combined femoropopliteal and tibial disease (HR 1.06, 95% CI: 1.05-1.08), iliac plus femoropopliteal disease (HR 1.06, 95% CI: 1.03–1.08), and multilevel disease (HR 1.17, 95% CI: 1.11-1.23) had a significantly higher risk of major amputation or death compared with those with isolated femoropopliteal disease (all p<0.0001). Importantly, the relative treatment effects of DCB±BMS and DES compared with PBA±BMS were consistent across all anatomical subgroups, with no significant interaction detected (all p for interaction>0.05). Treatment effects were consistent across rural and urban settings (interaction p=0.449). Among rural patients, the adjusted HR for DCB±BMS versus PBA±BMS was 0.95 (95% CI: 0.88-1.03) and for DES versus PBA±BMS, it was 0.94 (95% CI: 0.85-1.04), both of which were non-significant. In additional analyses using inverse probability of treatment weighting, baseline covariates were well balanced across treatment groups (all SMD <0.1), and weighted results were consistent with the main analysis: at 2 years, the adjusted hazard ratio for the composite outcome was 0.88 (95% CI: 0.86-0.90; p<0.0001) for DCB±BMS versus PBA±BMS, and 0.92 (95% CI: 0.89-0.94; p<0.0001) for DES versus PBA±BMS. At 5 years, results remained consistent (HR 0.91 and 0.94; both p<0.0001). In falsification endpoint analyses (pneumonia and hip fracture), adjusted cumulative incidences were similar across treatment groups (Supplementary Table 25).
Intravascular ultrasound (IVUS) use was identified from physician (Carrier) claims and was available for a subset of procedures. Among these, IVUS was coded in 884 of 20,878 procedures with PBA±BMS (4.2%), 796 of 18,127 with DCB±BMS (4.4%), and 831 of 9,129 with DES (9.1%)
Healthcare costs
Total healthcare costs varied substantially across device strategies. Cost values reflect cumulative expenditures from the index procedure to the end of follow-up. Compared with PBA±BMS, DES were associated with significantly higher overall Medicare payments (adjusted cost ratio 1.22, 95% CI: 1.21-1.24; p<0.0001) and higher total provider payments (adjusted cost ratio 1.19, 95% CI: 1.18-1.21; p<0.0001). In contrast, DCB±BMS was associated with no difference in Medicare payments (adjusted cost ratio 1.00, 95% CI: 0.99-1.01; p<0.5) and no significant difference in provider payments (adjusted cost ratio 0.99, 95% CI: 0.99-1.00; p=0.12). A visual summary of clinical and economic outcomes is presented in the Central illustration.
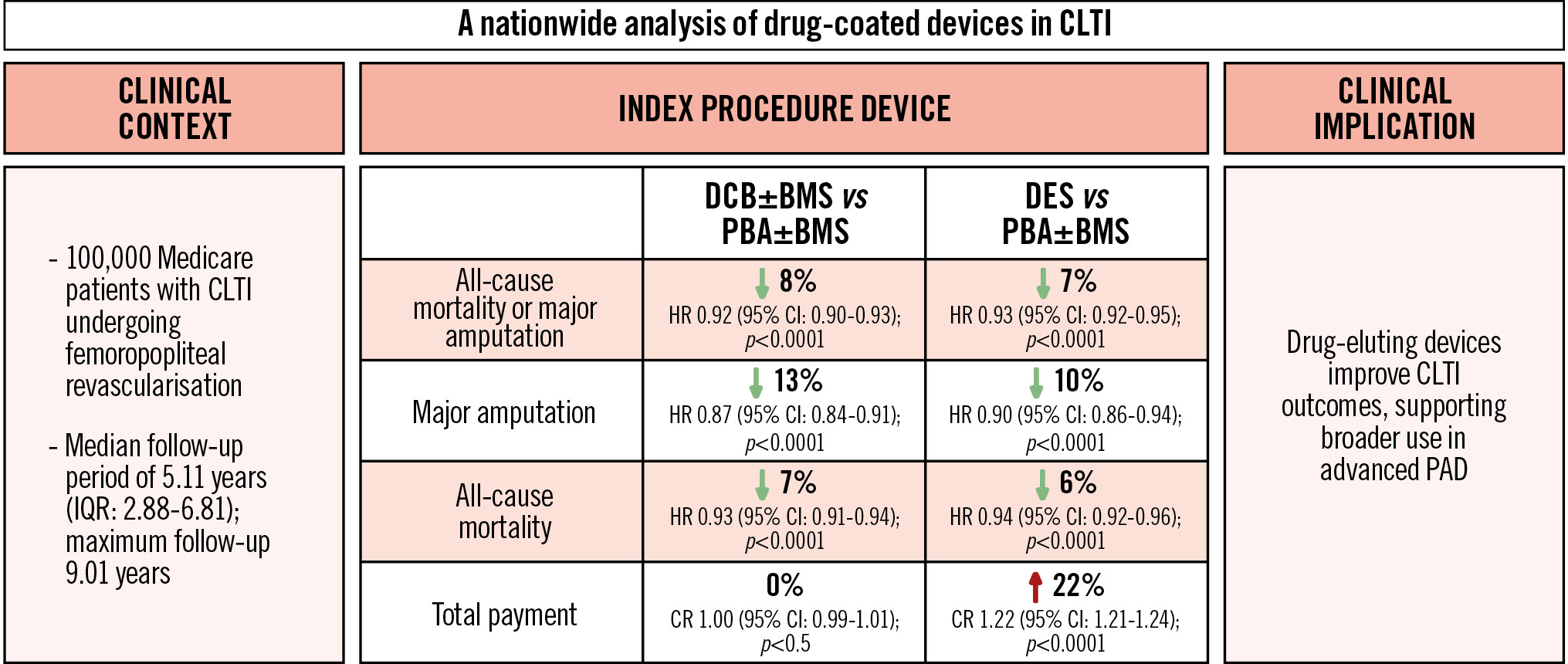
Central illustration. Comparative effectiveness of drug-coated devices for CLTI in a national Medicare cohort. This illustration summarises the comparative effectiveness and economic impact of drug-coated devices in over 100,000 Medicare patients with CLTI compared with treatment with PBA. DCBs and DES were both associated with a reduced risk of major amputation or death. DCBs also reduced reintervention rates and were cost-neutral, while DES showed similar clinical benefit but with increased healthcare costs. These findings support the use of drug-coated devices – particularly DCBs – in femoropopliteal revascularisation for CLTI and highlight the need for personalised, outcome-driven device selection. BMS: bare metal stent; CI: confidence interval; CLTI: chronic limb-threatening ischaemia; CR: cost ratio; DCB: drug-coated balloon; DES: drug-eluting stent; HR: hazard ratio; IQR: interquartile range; PAD: peripheral artery disease; PBA: plain balloon angioplasty
Discussion
Given the paucity of prospective data evaluating the role of drug-coated devices in CLTI revascularisation, our findings contribute important long-term, real-world evidence on the safety and clinical effectiveness of these devices. In this large national cohort of patients with CLTI undergoing FP revascularisation, we noted that many patients with CLTI required multilevel interventions, rather than only FP interventions. In this context, the use of drug-coated devices was associated with improved outcomes. These findings, consistent with the trends observed in BASIL-3, support the clinical value of drug-based therapies in high-risk CLTI patients. While secondary outcomes showed some divergence, with DCB±BMS demonstrating more consistent benefits on limb-related endpoints, both DCB±BMS and DES appear to offer meaningful improvements over uncoated devices for CLTI patients, which is also in line with recent guideline recommendations on the management of intermittent claudication21.
Compared with PBA±BMS, the use of DCB±BMS was associated with a 9% relative reduction in major amputation and a 2% reduction in reintervention. While absolute risk reductions were modest – approximately 2-3% at 2 years – this translated into a number needed to treat of 33 for major amputation and 27 for the composite outcome, reflecting a clinically meaningful impact in this high-risk population (Table 2, Figure 3). These findings align with previous randomised trials in patients with less severe disease, including IN.PACT SFA and LEVANT 2, where DCBs improved primary patency and freedom from target lesion revascularisation2223. While those trials primarily enrolled patients with claudication, our data extend the benefits of DCBs to a broader, sicker, and more diverse CLTI population. DCBs may be especially suitable for frail CLTI patients because of the absence of a permanent implant, reduced procedural time, and preserved surgical options2425. These advantages are reflected in real-world registries, such as the IN.PACT Global Study, which showed a durable benefit in complex anatomies26. Paclitaxel’s antiproliferative properties likely contribute to sustained patency and improved limb outcomes in CLTI. To confirm the robustness of these findings, we conducted a prespecified sensitivity analysis restricted to patients with available procedural laterality modifiers, allowing for precise attribution of amputations to the treated limb. In this rigorously defined subgroup, both DCBs and DES remained significantly associated with a lower risk of ipsilateral major amputation compared with PBA±BMS. These results strengthen the internal validity of our findings and mitigate concerns about potential misclassification or outcome dilution related to non-lateralised events.
In contrast, DES were associated with significant reductions in both all-cause mortality and major amputation but did not meaningfully reduce MALE rates compared with PBA±BMS. This disconnect between amputation-free survival and reintervention rates suggests benefits beyond target lesion patency. Notably, DES-treated patients incurred the highest costs across all treatment strategies, which could be driven by a greater use of combined therapies, such as DCBs with DES. These results should be interpreted in the context of prior randomised trials conducted in patients with intermittent claudication, where DES have consistently demonstrated improved patency and a reduced need for reintervention. For instance, in the EMINENT and SPORTS trials, the Eluvia DES (Boston Scientific) showed superior patency and reduced reinterventions compared with BMS and DCBs27. While the EMINENT trial exclusively enrolled claudicants, the SPORTS trial included a minority of patients with ischaemic rest pain (Rutherford category 4). However, both trials underrepresented the full clinical spectrum of CLTI, especially patients with tissue loss, which underscores the importance of our study’s focus on a real-world CLTI population.
Notably, our results align in part with the BASIL-3 trial, which showed numerical trends, albeit non-significant, towards improvement in amputation-free survival with drug-coated devices9. Our results are also consistent with prior large observational studies such as SAFE-PAD, which found no excess mortality associated with paclitaxel exposure over long-term follow-up among both claudicants and patients with CLTI, and the SFA-Long study, which reported durable benefits of drug-coated balloons in femoropopliteal disease, including in patients with CLTI, with improved amputation-free survival at 5 years1128. With a substantially larger sample size and follow-up period, our analysis yielded similar absolute risk differences to those of BASIL-3, but with sufficient power to meet statistical significance. Several key distinctions are critical for comparing the results across these studies. The BASIL-3 population had milder disease, with fewer cases of gangrene and comorbidities, and the results were affected by crossover. Our study included a sicker population and longer follow-up, providing complementary real-world insight.
Interestingly, a recent exploratory analysis of the BEST-CLI trial presented at the 2025 Society for Vascular Surgery meeting by Siracuse et al further supports the potential benefit of drug-coated technologies. In this secondary analysis of patients undergoing femoropopliteal endovascular interventions, the use of drug-coated devices was associated with significantly lower mortality (HR 0.53, 95% CI: 0.31-0.91; p=0.02) and fewer major reinterventions (HR 0.57, 95% CI: 0.31-0.91; p=0.02) compared with non-drug devices29. Although derived from a small and selected subset of BEST-CLI patients, this analysis supports the hypothesis that fewer limb events may translate into improved survival. While these findings remain exploratory, they echo the signals observed in our Medicare cohort and lend further support to the clinical relevance of drug-coated endovascular strategies.
Another important consideration is the economic footprint of drug-based technologies. The adoption of drug-coated devices in femoropopliteal revascularisations has fluctuated over recent years, reflecting evolving clinical confidence and emerging safety data, particularly concerning their use in the CLTI population3031. This trend likely reflects favourable patency, low complication rates, and growing clinical familiarity with drug-delivery technologies. While this expansion may contribute to higher procedural costs, drug-coated devices still represent a rational investment if these devices provide even modest benefits in outcomes such as wound healing or freedom from reintervention – benefits that can be particularly meaningful in patients with advanced disease. In our study, we observed that most of the clinical benefit with drug-coated devices emerged within the first 24 months, suggesting an early separation of event curves. This temporal profile helps address concerns regarding delayed harm and reinforces the short- to midterm value of these devices in the high-risk CLTI population. This is counterbalanced by the extreme mortality risk of the CLTI population, as highlighted by the high rates of death observed in this analysis. Interestingly, the BASIL-3 trial also included a formal cost-effectiveness analysis, which demonstrated that DES achieved financial superiority over PBA±BMS, while DCBs showed borderline cost-effectiveness, further supporting the value of DES in selected patients9. Beyond cost-effectiveness, recent randomised evidence has also provided important reassurance regarding the safety of paclitaxel-coated devices in CLTI. In both the BASIL-3 trial and the SWEDEPAD 2 trial, no excess mortality was observed with paclitaxel-coated balloons or stents compared with uncoated technologies, even with extended follow-up932. These findings are further supported by the SAFE-PAD study, which specifically included patients with CLTI and confirmed the absence of an association between paclitaxel dose and long-term mortality1133. Together, these findings mitigate the earlier concerns of dose-dependent toxicity raised by post hoc meta-analyses and support the long-term safety of drug-coated devices in real-world CLTI practice.
Table 2. Primary composite and individual outcomes with absolute risk differences and number needed to treat.
| Comparison | Endpoint | ARD at 2 years | NNT (2 years) | ARD at 5 years | NNT (5 years) |
|---|---|---|---|---|---|
| DCB±BMS vs PBA±BMS | Composite* (death or amputation) | 7.46% | 13 | 4.50% | 22 |
| DCB±BMS vs PBA±BMS | Major amputation | 2.51% | 40 | 2.24% | 45 |
| DES vs PBA±BMS | Composite | 6.83% | 15 | 3.98% | 25 |
| DES vs PBA±BMS | Major amputation | 2.33% | 43 | 2.01% | 50 |
| *Composite: major amputation, death (all-cause), or reintervention. ARD: absolute risk difference; BMS: bare metal stent; DCB: drug-coated balloon; DES: drug-eluting stent; NNT: number needed to treat; PBA: plain balloon angioplasty | |||||
Limitations
This study has important limitations. The observational design and use of administrative data may introduce residual confounding. Furthermore, claims-based data have limited granular detail regarding anatomical and procedural characteristics, such as lesion length, calcification severity, and runoff status. In addition, this study was unable to capture patient-reported outcomes, quality of life, or limb function – all increasingly important in the CLTI population. Although we adjusted for disease severity, comorbidities, and hospital-level characteristics, residual confounding by indication cannot be fully excluded. In this context, contemporary US guidelines recommend the use of drug-coated devices across the spectrum of CLTI presentations, not restricted to specific patient subsets; this mitigates the concern that these technologies were selectively used in more favourable anatomies34. Finally, we were unable to directly assess index lesion patency with the different treatment modalities, as angiographic or duplex ultrasound-based patency data are not available in the Medicare claims database. However, we reported clinically relevant endpoints – major amputation, mortality, reintervention, and healthcare utilisation – that serve as indirect surrogates of device performance in this real-world population. Because our study cohort consisted exclusively of older US Medicare beneficiaries, the generalisability of our findings to younger patients or to health systems outside the United States remains limited. However, this study also has key strengths, including a large, diverse, and representative national cohort of patients; complete follow-up ascertainment; clinically relevant endpoints; and robust subgroup and temporal analyses – all of which make the findings highly generalisable to real-world CLTI care.
Conclusions
In this national cohort of over 100,000 real-world patients undergoing FP revascularisation for CLTI, drug-coated devices were associated with modest but consistent reductions in major amputation or death compared to uncoated devices. These findings differ from the recent BASIL-3 randomised trial, which showed similar numerical trends towards benefit with drug-coated devices but failed to reach statistical significance in any comparison. In addition to the need for more prospective randomised trial data, clinical decision-making in CLTI should continue to prioritise outcome-driven, cost-conscious strategies informed by both randomised and real-world evidence.
Impact on daily practice
This nationwide analysis supports a paradigm shift towards the routine use of drug-coated balloons (DCBs) in femoropopliteal revascularisation for patients with chronic limb-threatening ischaemia. In a high-risk Medicare population, DCBs significantly reduced major amputation, mortality, and reintervention, without increasing overall costs. These findings endorse DCBs as a first-line endovascular strategy, combining clinical efficacy with economic efficiency. In daily practice, DCBs offer a compelling option to optimise outcomes in frail patients, limit the use of permanent implants, and preserve future revascularisation strategies, thus aligning with the principles of durable, patient-centred limb salvage.
Conflict of interest statement
J. Sobocinski declares consulting fees for Abbott and W. L. Gore & Associates. J.M. Kim is supported by the NIH T32 HL160522 grant. Y. Gouëffic reports research funding from PrediSurge, Biotronik, W. L. Gore & Associates, and GE HealthCare; and honoraria from Abbott, BD, Bentley, Biotronik, Boston Scientific, Cook Medical, Eclevar Medtech, GE HealthCare, iVascular, Medtronic, Penumbra, Sensome, Shockwave Medical, and W. L. Gore & Associates (consulting, medical advisory board, educational course, speaking). O. Iida declares: consulting for Abbott, Boston Scientific, Gore Medical, Kaneka Medix, Tokai Medical, OrbusNeich, and Otsuka Medical; and honoraria from Boston Scientific, Gore Medical, Kaneka Medix, Tokai Medical, OrbusNeich, Otsuka Medical, Medtronic, Terumo, Nipro, and Canon. E.A. Secemsky receives funding from NIH/NHLBI K23HL150290 and the U.S. Food & Drug Administration; has institutional grants from Abbott/CSI, BD, Boston Scientific, Cook Medical, Medtronic, and Philips; and receives speaking/consulting fees from Abbott/CSI, BD, BMS, Boston Scientific, Cagent, Conavi, Cook Medical, Cordis, Endovascular Engineering, W. L. Gore & Associates, InfraRedx, Medtronic, Philips, RapidAI, Rampart, Shockwave Medical, Siemens, Teleflex, Terumo, Thrombolex, VentureMed, and Zoll. The other authors have no conflicts of interest to declare.
Supplementary data
To read the full content of this article, please download the PDF.

