Cory:
Unlock Your AI Assistant Now!
A 73-year-old female with a history of rheumatic fever and severe mitral stenosis who had undergone a valve-in-mitral annular calcification (ViMAC) transcatheter mitral valve replacement (TMVR) with a 26 mm SAPIEN 3 Ultra valve (Edwards Lifesciences) 5 years prior, presented with New York Heart Association (NYHA) Class III symptoms. Transoesophageal echocardiography (TOE) demonstrated a well-functioning mitral transcatheter heart valve (THV) (Figure 1A) with mild paravalvular leak (PVL) (Figure 1B). Transthoracic echocardiography (TTE), however, revealed severe tricuspid regurgitation (TR) (Figure 1C). TOE imaging of the tricuspid valve was markedly impaired due to acoustic shadowing from the mitral THV frame and calcified annulus, which severely limited leaflet visualisation (Figure 1D, Figure 1E, Moving image 1, Moving image 2).
Given the high surgical risk, the Heart Team proceeded with transcatheter tricuspid valve replacement (TTVR) with a 44 mm EVOQUE valve (Edwards Lifesciences) via right femoral venous access under general anaesthesia. Despite the limited TOE imaging (Figure 1F, Figure 1G, Moving image 3), a three-dimensional intracardiac echocardiography (3D ICE) catheter (VeriSight Pro [Philips]) positioned in the right atrium immediately posterior to the tricuspid valve provided excellent visualisation of the tricuspid leaflets, free from acoustic interference by the mitral THV or MAC. The valve deployment was guided primarily by 3D ICE (Figure 1H, Figure 1I, Moving image 4) and fluoroscopy (right anterior oblique 18°, caudal 1° projection).
The postprocedural course was uneventful. TTE on postoperative day 1 showed a well-expanded prosthesis with a mean gradient of 5 mmHg and no PVL (Figure 1J). At 30 days, the patient improved to NYHA Class I. TTE confirmed trivial TR, no PVL, and a mean gradient of 7 mmHg, while computed tomography confirmed well-seated atrioventricular THVs (Figure 1K, Figure 1L, Moving image 5).
This case highlights the feasibility of TTVR after ViMAC TMVR and underscores the pivotal role of 3D ICE in guiding tricuspid interventions when TOE visualisation is limited by MAC and a mitral prosthesis1.
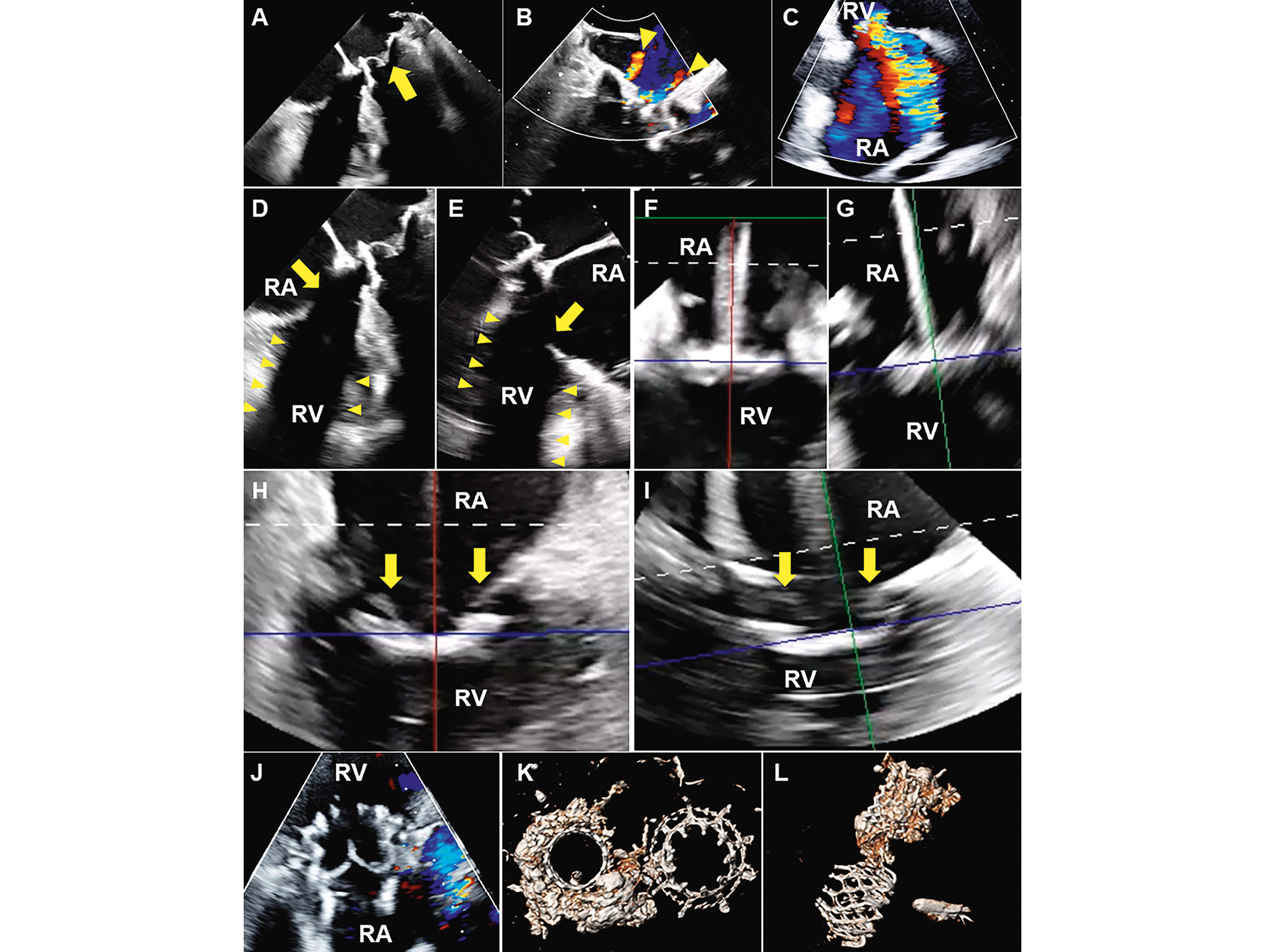
Figure 1. 3D ICE-guided TTVR 5 years after TMVR. A,B) Preprocedural TOE showed a well-functioning SAPIEN 3 Ultra mitral prosthesis (yellow arrow) with two small paravalvular leak jets (yellow arrowheads). C) Preprocedural TTE revealed severe tricuspid regurgitation. D,E) Pre- and (F,G) intraprocedural TOE demonstrated tricuspid leaflets (yellow arrows) obscured by acoustic shadowing from the mitral transcatheter heart valve (yellow arrowheads). H,I) Three-dimensional intracardiac echocardiography clearly visualised tricuspid leaflets (yellow arrows). At 30 days, (J) TTE demonstrated no tricuspid regurgitation or paravalvular leak; and (K,L) computed tomography confirmed well-expanded transcatheter mitral and tricuspid prostheses and severe mitral annular calcification. 3D ICE: three-dimensional intracardiac echocardiography; RA: right atrium; RV: right ventricle; TOE: transoesophageal echocardiography; TTE: transthoracic echocardiography; TMVR: transcatheter mitral valve replacement; TTVR: transcatheter tricuspid valve replacement
Conflict of interest statement
S. Khera is a consultant and proctor for Medtronic and Abbott; a consultant for Terumo and W. L. Gore & Associates; a consultant and advisory board member of East End Medical; and serves on the speaker’s bureau for Zoll Medical and Edwards Lifesciences. L.M. Safi serves on the speaker’s bureau for Abbott and Medtronic. G.H.L. Tang has received speaker honoraria and served as a physician proctor, consultant, advisory board member, TAVR publications committee member, RESTORE study steering committee member, APOLLO trial screening committee member, and IMPACT MR steering committee member for Medtronic; has received speaker honoraria and served as a physician proctor, consultant, advisory board member, ENVISION trial screening committee member, and TRILUMINATE trial anatomic eligibility and publications committee member for Abbott; has served as an advisory board member for Boston Scientific; a consultant and physician screening committee member for Shockwave Medical; a consultant for Philips, Edwards Lifesciences, Peija Medical, and Shenqi Medical Technology; and has received speaker honoraria from Siemens Healthineers. The other authors have no relevant conflicts of interest to declare.
Supplementary data
To read the full content of this article, please download the PDF.
Moving image 4. Three-dimensional intracardiac echocardiogram.
Moving image 5. 30-day transthoracic echocardiogram.
Moving image 1. Preprocedural transoesophageal echocardiogram, part 1.
Moving image 2. Preprocedural transoesophageal echocardiogram, part 2.
Moving image 3. Intraprocedural transoesophageal echocardiogram.

